Discovery of first-in-class human glycolate oxidase near infrared molecular rotor inhibitors (NIRGOi)
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
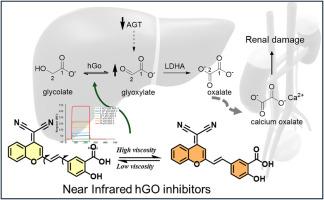
Primary hyperoxaluria type 1 (PH1) is a rare genetic disorder that affects the metabolism of oxalate leading to formation of renal calculi and end stage kidney failure. Currently, no specific drug exists for PH1 treatment, while human glycolate oxidase (hGO), which influences endogenous oxalate synthesis, is a clinically validated target for PH1 treatment. We envisioned the possibility of exploiting the reported salicylate scaffold as an hGO interactor to develop the first in their class Near Infrared hGO inhibitors, that we termed NIRGOi. These could enable the simultaneous tracking and inhibition of hGO in a single moiety. We encompassed different electron acceptors (A) and 5-formylsalicylic acid, the electron donor (D), as a two in one moiety, to develop three D-A-type NIRGOi, new compounds capable of enhancing their photophysical properties upon interaction with the target protein, hGO. Their photophysical properties verified that the D–A interactions successfully redshifted the emission maxima to wavelengths ranging from 550 nm to 690 nm. Their interaction with hGO was first modelled in silico and then an array of in vitro assays were used to verify their low micromolar efficacy along with the alteration of their photophysical properties upon binding. This study provides new tools, the NIRGOi, that are promising starting points for the development of NIR reporting probes to explore and potentially treat Primary Hyperoxaluria type 1.

求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


