Mass transfer modeling of absorption and desorption behavior of atmospheric water with an aqueous potassium acetate solution in a packed column
IF 4.1
2区 工程技术
Q2 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
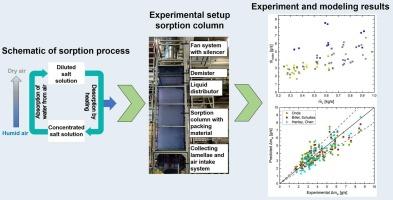
The potential of absorptive atmospheric water generation using a liquid salt solution offers the advantage of good scalability and specific uptake potential for atmospheric water. To investigate the sorption kinetics of an aqueous potassium acetate solution, a packed column experimental setup was constructed. A total of 99 runs were conducted to determine the sorption kinetics and the impact of varying experimental parameters. A maximum absorption rate of up to 8.6 g/s from the air was achieved with the mass flow rates and the inlet air humidity having the greatest positive impact on the absorption rate. Furthermore, the mass transfer was modeled using three distinct models for the mass transfer coefficients and the effective mass transfer area. The findings indicate that the Billet-Schultes model exhibits the least deviation from the observed results with an average of 15.6 % for absorption. The Onda (22.7 %) and Hanley-Chen model (16.8 %) demonstrate comparatively higher average deviations.

求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Engineering Science
工程技术-工程:化工
CiteScore
7.50
自引率
8.50%
发文量
1025
审稿时长
50 days
期刊介绍:
Chemical engineering enables the transformation of natural resources and energy into useful products for society. It draws on and applies natural sciences, mathematics and economics, and has developed fundamental engineering science that underpins the discipline.
Chemical Engineering Science (CES) has been publishing papers on the fundamentals of chemical engineering since 1951. CES is the platform where the most significant advances in the discipline have ever since been published. Chemical Engineering Science has accompanied and sustained chemical engineering through its development into the vibrant and broad scientific discipline it is today.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


