Exploring chloro-isoxazole compounds inspired on tetrahydrofuran neolignans as promising antileishmanial agents
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
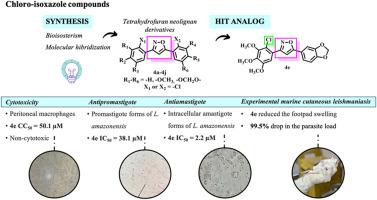
The present work aims to synthesize and to identify a potential antileishmanial agent from chloro-diphenyl isoxazole methoxylated compounds. We have synthesized ten new chloro-isoxazole analogs inspired by the scaffold of tetrahydrofuran neolignans veraguensin 1a, grandisin 1b, and machilin G 1c. To obtain analogs 4a-4j, we used a cycloaddition reaction with yields ranging from 45 % to 89 %. All compounds were characterized using Nuclear Magnetic Resonance of 1H and 13C and analyzed by using High-Resolution Mass Spectrometry. The HPLC analysis confirmed that all compounds were more than 95 % pure. Finally, we tested the antileishmanial activity of these analogs against promastigote and intracellular amastigote forms of L. amazonensis in vitro. We conducted tests on murine peritoneal macrophages to determine the cytotoxicity of the analogs. Our findings revealed that 4e (R1–R3 = –OCH3, X1 = -Cl, R4 and R5 = –OCH2O-), a hybrid compound of grandisin and machilin G, showed moderate activity on promastigotes (IC50 = 38.1 ± 1.5 μM). 4e was also effective against intracellular amastigotes with similar IC50 values to AmB-treated control (IC50 = 2.2 ± 0.4 μM and IC50 = 2.0 ± 0.1, respectively). Moreover, it exhibited a selectivity index (SI) for amastigote forms equal to 22.7, higher than the reference drugs we tested. Analog 4e displayed non-mutagenic potential at all tested concentrations in the Ames test. We also evaluated the therapeutic effect of 4e on the experimental cutaneous leishmaniasis model with BALB/c mice infected with promastigote forms of L. amazonensis and treated with intralesional (IL) injections. Our study found that mice treated with 4e had a significant reduction (99.5 % drop) in the footpad tissue parasite load compared to the control group treated with the vehicle. The effect of 4e was similar, controlling the infection, to that of N-methylglucamine antimonate (Sb, Glucantime, 99.8 % drop), which is a reference treatment. Based on our results, we suggest that chloro-isoxazole analog 4e shows potential as an antileishmanial agent for treating cutaneous leishmaniasis (CL).

探索以四氢呋喃新木质素为灵感的氯代异噁唑化合物作为有前途的抗利什曼病药物
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


