Design, synthesis, and biological evaluation of 4-(2-fluorophenoxy)pyridine derivatives as novel FLT3-ITD inhibitors
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
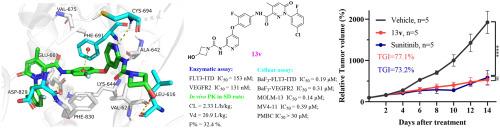
FMS-like tyrosine kinase 3 (FLT3) is an ideal drug target for the treatment of acute myeloid leukemia (AML). Although several FLT3 inhibitors have been approved or evaluated in clinical trials, selectivity over c-Kit kinase and FLT3 WT remains a major challenge. Herein, we report a series of 4-(2-fluorophenoxy)pyridine derivatives with potent inhibitory activities against FLT3 internal tandem duplication (FLT3-ITD). The representative compound 13v inhibited FLT3-ITD kinase and isogenic BaF3-FLT3-ITD cells with nanomolar IC50 values and achieved selectivity over c-Kit (>53-fold) and FLT WT (19-fold) in transformed BaF3 cells. In addition, compound 13v displayed excellent selectivity against FLT3-ITD driven AML cells compared to other leukemia cells, solid tumors, and normal peripheral blood mononuclear cells. Mechanistic studies revealed that 13v disrupted FLT3 signal transduction and induced G0/G1 cell cycle arrest and apoptosis. Moreover, it also showed good developmental profiles in ADME assays. In in vivo studies, 13v demonstrated desirable pharmacokinetic (PK) profiles and sufficient tumor growth inhibition in a MOLM-13 xenograft model. Taken together, 13v may represent a starting point for the development of improved FLT3-ITD inhibitors.

作为新型 FLT3-ITD 抑制剂的 4-(2-氟苯氧基)吡啶衍生物的设计、合成和生物学评价
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


