Deciphering the Antibacterial Mechanism of Hollow Sphere Metal–Organic Frameworks against Staphylococcus aureus and Its Application in Wound Healing
IF 7.2
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
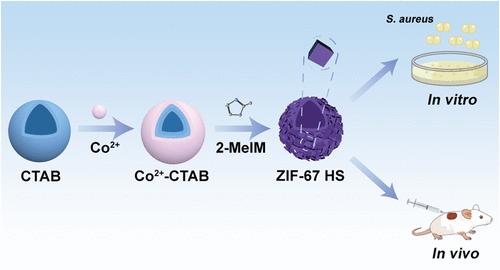
Staphylococcus aureus infection and antimicrobial resistance (AMR) to antibiotics are two significant factors that pose escalating threats to public health, potentially leading to persistent pain or even death. Consequently, there is an urgent need for the development of alternative antibacterial materials. In this study, we have successfully developed a biocompatible nanosystem based on hollow spherical zeolitic imidazolate framework-67 (ZIF-67 HS), which exhibits remarkable bactericidal efficacy against Gram-positive S. aureus both in vitro and in vivo. Importantly, our findings demonstrate that prolonged exposure of S. aureus to subminimum inhibitory concentrations of ZIF-67 HS does not induce secondary resistance as observed with conventional antibiotics; instead, it maintains sustained susceptibility toward antibiotic-resistant strains. Multiomics analyses (including transcriptomics, metabolomics, and metalomics) have indicated that ZIF-67 HS regulates 95 genes, 261 metabolites, and 2 metal elements in S. aureus. Through bioinformatics analysis in combination with bioassays, we demonstrated that ZIF-67 HS disrupted multiple essential pathways in the pathogen, including amino acid metabolism, iron metabolism, membrane transport, reactive oxygen species (ROS), and S. aureus infection pathways. Our findings suggest that ZIF-67 HS induces bacterial death through multiple mechanisms rather than a single target and highlight its long-lasting antibacterial properties. This study presents valuable insights into the potential of nanobiomedicine for eradicating bacteria and elucidates the related molecular mechanism.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemistry of Materials
工程技术-材料科学:综合
CiteScore
14.10
自引率
5.80%
发文量
929
审稿时长
1.5 months
期刊介绍:
The journal Chemistry of Materials focuses on publishing original research at the intersection of materials science and chemistry. The studies published in the journal involve chemistry as a prominent component and explore topics such as the design, synthesis, characterization, processing, understanding, and application of functional or potentially functional materials. The journal covers various areas of interest, including inorganic and organic solid-state chemistry, nanomaterials, biomaterials, thin films and polymers, and composite/hybrid materials. The journal particularly seeks papers that highlight the creation or development of innovative materials with novel optical, electrical, magnetic, catalytic, or mechanical properties. It is essential that manuscripts on these topics have a primary focus on the chemistry of materials and represent a significant advancement compared to prior research. Before external reviews are sought, submitted manuscripts undergo a review process by a minimum of two editors to ensure their appropriateness for the journal and the presence of sufficient evidence of a significant advance that will be of broad interest to the materials chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


