Synthesis and multiple responsiveness study of Poly(N-isopropylacrylamide) containing azopyridine
IF 4.1
2区 化学
Q2 POLYMER SCIENCE
引用次数: 0
Abstract
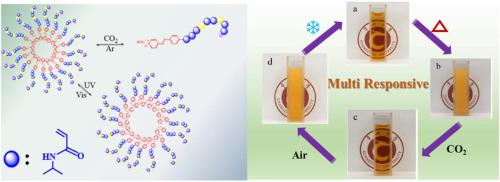
Environmentally sensitive polymer materials exhibit diverse properties due to their ability to respond to external environmental changes such as light, temperature, pH, and et al. These responses lead to corresponding alterations in their structure or state, resulting in unexpected performance. In this study, azo pyridine groups were introduced into the end groups and side chains of PNIPAM to synthesize two distinct polymer structures (PNIPAM-AZO and P (NIPAM-co-PAZO-co-OEGMA). This integration enables the polymers to respond to multiple stimuli, including temperature, light, pH, and gas (CO2). A comprehensive investigation of the properties of these multi-responsive polymer materials was conducted. The findings reveal that the presence of azo pyridine units enables both polymers to modulate the (lower critical solution temperature) LCST of PNIPAM through UV, pH, and CO2, with a maximum temperature adjustment of 13 °C when CO2 is introduced. This modulation range is influenced by the azo pyridine content in the polymer. Notably, when azo pyridine is present only at the polymer end group, the maximum LCST regulation range is limited to 3 °C. Moreover, the LCST of the two polymers can also be adjusted by pH. In the presence of carbonate ions, the protonation of azo pyridine by carbonic acid increases the solubility of the polymer and enhances the LCST. Introducing argon gas removes CO2, thereby restoring the polymer's solubility and LCST to their neutral condition levels. By alternately introducing CO2 and argon gas, the LCST of the polymer exhibits cyclic changes.

求助全文
约1分钟内获得全文
求助全文
来源期刊

Polymer
化学-高分子科学
CiteScore
7.90
自引率
8.70%
发文量
959
审稿时长
32 days
期刊介绍:
Polymer is an interdisciplinary journal dedicated to publishing innovative and significant advances in Polymer Physics, Chemistry and Technology. We welcome submissions on polymer hybrids, nanocomposites, characterisation and self-assembly. Polymer also publishes work on the technological application of polymers in energy and optoelectronics.
The main scope is covered but not limited to the following core areas:
Polymer Materials
Nanocomposites and hybrid nanomaterials
Polymer blends, films, fibres, networks and porous materials
Physical Characterization
Characterisation, modelling and simulation* of molecular and materials properties in bulk, solution, and thin films
Polymer Engineering
Advanced multiscale processing methods
Polymer Synthesis, Modification and Self-assembly
Including designer polymer architectures, mechanisms and kinetics, and supramolecular polymerization
Technological Applications
Polymers for energy generation and storage
Polymer membranes for separation technology
Polymers for opto- and microelectronics.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


