Cracking melanoma’s armor: Supramolecular dual-strike on XPO1 and β-catenin to overcome resistance
IF 11.4
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
Introduction
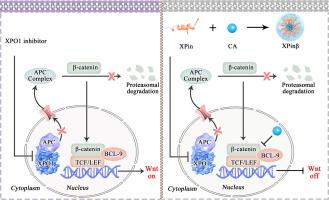
XPO1 plays a crucial role in the nuclear export machinery, making it an attractive target for inhibiting nuclear-cytoplasmic transport in melanoma, where its overexpression is linked to unfavorable prognosis. However, XPO1 monotherapy has not demonstrated sufficient efficacy to be considered a first-line treatment option for melanoma.Objectives
This research aimed to delve into the resistance mechanism of XPO1-targeting therapy in melanoma and fabricate a proteinoid microsphere which could target XPO1 and β-catenin to maximize the effect of XPO1 inhibitors.Methods
Transcriptome sequencing was used to analyze the effects of XPO1 interference on the signaling pathways of melanoma. Nuclear-cytoplasmic protein separation, co-immunoprecipitation, and confocal microscopic analyses were conducted to clarify the resistance mechanism of XPO1 targeting therapy. A proteinoid microsphere named XPinβ was developed by co-assembling a specially designed XPO1 antagonistic peptide (XPin) and a β-catenin antagonist (Carnosic acid/CA). Cell model, mouse allograft and patient-derived xenograft (PDX) models were used to evaluate the antitumor effect of XPinβ.Results
In our study, inhibition of XPO1 led to the nuclear accumulation of β-catenin, altered the nuclear-cytoplasmic localization of APC, and activated the Wnt/β-catenin signaling pathway. XPinβ was efficiently internalized into melanoma cells via macropinocytosis, achieving simultaneous inhibition of both XPO1 and β-catenin. As expected, XPinβ demonstrated robust anti-tumor efficacy in an allograft melanoma mouse model, with significantly superior therapeutic effects compared to monotherapy targeting XPO1 or CA treatment alone. Moreover, XPinβ effectively inhibited growth of patient-derived xenograft (PDX) tumors overexpressing XPO1, outperforming both CA and the commercially available XPO1 inhibitor KPT-330. Most importantly, XPinβ significantly suppressed pulmonary metastasis of melanoma while maintaining excellent biosafety.Conclusions
This study demonstrates the enhanced efficacy of XPO1-targeted therapy through the inhibition of the Wnt/β-catenin signaling pathway and introduces XPinβ, a proteinoid microsphere with promising clinical translational potential for dual targeting therapy against melanoma involving both XPO1 and β-catenin.
破解黑色素瘤的盔甲:超分子双重打击 XPO1 和 β-catenin,克服抗药性
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Advanced Research
Multidisciplinary-Multidisciplinary
CiteScore
21.60
自引率
0.90%
发文量
280
审稿时长
12 weeks
期刊介绍:
Journal of Advanced Research (J. Adv. Res.) is an applied/natural sciences, peer-reviewed journal that focuses on interdisciplinary research. The journal aims to contribute to applied research and knowledge worldwide through the publication of original and high-quality research articles in the fields of Medicine, Pharmaceutical Sciences, Dentistry, Physical Therapy, Veterinary Medicine, and Basic and Biological Sciences.
The following abstracting and indexing services cover the Journal of Advanced Research: PubMed/Medline, Essential Science Indicators, Web of Science, Scopus, PubMed Central, PubMed, Science Citation Index Expanded, Directory of Open Access Journals (DOAJ), and INSPEC.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


