Guidelines for minimal reporting requirements, design and interpretation of experiments involving the use of eukaryotic dual gene expression reporters (MINDR)
IF 10.1
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
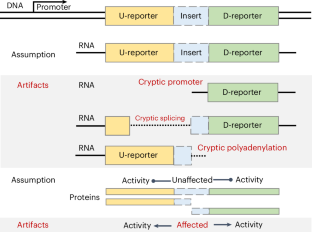
Dual reporters encoding two distinct proteins within the same mRNA have had a crucial role in identifying and characterizing unconventional mechanisms of eukaryotic translation. These mechanisms include initiation via internal ribosomal entry sites (IRESs), ribosomal frameshifting, stop codon readthrough and reinitiation. This design enables the expression of one reporter to be influenced by the specific mechanism under investigation, while the other reporter serves as an internal control. However, challenges arise when intervening test sequences are placed between these two reporters. Such sequences can inadvertently impact the expression or function of either reporter, independent of translation-related changes, potentially biasing the results. These effects may occur due to cryptic regulatory elements inducing or affecting transcription initiation, splicing, polyadenylation and antisense transcription as well as unpredictable effects of the translated test sequences on the stability and activity of the reporters. Unfortunately, these unintended effects may lead to misinterpretation of data and the publication of incorrect conclusions in the scientific literature. To address this issue and to assist the scientific community in accurately interpreting dual-reporter experiments, we have developed comprehensive guidelines. These guidelines cover experimental design, interpretation and the minimal requirements for reporting results. They are designed to aid researchers conducting these experiments as well as reviewers, editors and other investigators who seek to evaluate published data. Here, the authors discuss dual-reporter systems and confounding variables that may lead to misinterpretation of results. They propose guidelines for robust design, physiological interpretation and accurate reporting when using dual reporters.

涉及使用真核双基因表达报告器(MINDR)的实验的最低报告要求、设计和解释指南
双报告者在同一mRNA中编码两种不同的蛋白质,在鉴定和表征真核生物翻译的非常规机制中起着至关重要的作用。这些机制包括通过内部核糖体进入位点(IRESs)起始,核糖体移框,停止密码子读取和再起始。这种设计使一个记者的表达受到被调查的特定机制的影响,而另一个记者则起到内部控制的作用。然而,当在这两个报告者之间放置干预测试序列时,挑战就出现了。这样的序列可能会不经意地影响两个报告基因的表达或功能,而不受翻译相关变化的影响,从而潜在地影响结果。这些影响可能是由于隐调控元件诱导或影响转录起始、剪接、聚腺苷酸化和反义转录,以及翻译的测试序列对报告子的稳定性和活性的不可预测的影响。不幸的是,这些意想不到的影响可能导致对数据的误解,并在科学文献中发表错误的结论。为了解决这个问题,并协助科学界准确解释双报告者实验,我们制定了全面的指导方针。这些准则涵盖实验设计、解释和报告结果的最低要求。它们旨在帮助进行这些实验的研究人员,以及寻求评估已发表数据的审稿人、编辑和其他调查人员。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


