Improving Solid-State Properties of the Side-Chain Peptide Building Blocks for the Synthesis of GLP-1 Analogs through Their Complexation with Metal Cations
IF 3.1
3区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
Abstract
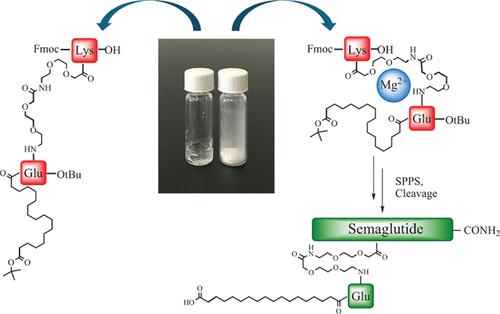
Glucagon-like peptide-1 analogs are receiving exponential attention for the treatment of type II diabetes and controlling weight due to their unique structure with non-natural lipidated side chains. However, the industrial-scale manufacturing of such peptides poses noticeable challenges due to the gel-like appearance of the peptide intermediates used to form the branched structure. Here, we found an efficient approach for the complexation of these protected peptide building blocks with magnesium chloride, which allows for obtaining free-flowing stable solids suitable for the more process-friendly synthesis of the peptides. A screening of the solvents and magnesium chloride content allowed us to determine the optimal parameters for the preparation of the complexes. NMR spectral analysis showed the involvement of amide groups in the interaction with the magnesium salt. Lastly, the efficiency of the coupling of the Mg-complexed lipidated side-chain building blocks was confirmed for the synthesis of tirzepatide and semaglutide fragments, showing a comparable performance with respect to the noncomplexed derivatives.
通过金属阳离子络合改善GLP-1类似物合成的侧链肽构建块的固态性能
胰高血糖素样肽-1 类似物因其具有非天然脂化侧链的独特结构,在治疗 II 型糖尿病和控制体重方面正受到越来越多的关注。然而,由于用于形成支链结构的肽中间体外观呈凝胶状,工业化生产此类肽带来了明显的挑战。在这里,我们发现了一种将这些受保护的肽结构单元与氯化镁复配的有效方法,这种方法可以获得自由流动的稳定固体,适用于工艺更友好的肽合成。通过对溶剂和氯化镁含量的筛选,我们确定了制备复合物的最佳参数。核磁共振光谱分析显示,酰胺基团参与了与镁盐的相互作用。最后,在合成替扎帕肽和塞马鲁肽片段时,镁络合脂化侧链构件的偶联效率得到了证实,与非络合衍生物相比,其性能相当。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
6.90
自引率
14.70%
发文量
251
审稿时长
2 months
期刊介绍:
The journal Organic Process Research & Development serves as a communication tool between industrial chemists and chemists working in universities and research institutes. As such, it reports original work from the broad field of industrial process chemistry but also presents academic results that are relevant, or potentially relevant, to industrial applications. Process chemistry is the science that enables the safe, environmentally benign and ultimately economical manufacturing of organic compounds that are required in larger amounts to help address the needs of society. Consequently, the Journal encompasses every aspect of organic chemistry, including all aspects of catalysis, synthetic methodology development and synthetic strategy exploration, but also includes aspects from analytical and solid-state chemistry and chemical engineering, such as work-up tools,process safety, or flow-chemistry. The goal of development and optimization of chemical reactions and processes is their transfer to a larger scale; original work describing such studies and the actual implementation on scale is highly relevant to the journal. However, studies on new developments from either industry, research institutes or academia that have not yet been demonstrated on scale, but where an industrial utility can be expected and where the study has addressed important prerequisites for a scale-up and has given confidence into the reliability and practicality of the chemistry, also serve the mission of OPR&D as a communication tool between the different contributors to the field.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


