MicroRNA-induced reprogramming of tumor-associated macrophages for modulation of tumor immune microenvironment
IF 10.5
1区 医学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
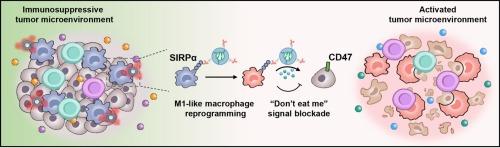
Tumor-associated macrophages (TAMs) are abundant in the tumor microenvironment and typically exhibit pro-tumoral phenotypes. TAMs overexpress the signal regulatory protein alpha (SIRPα) receptor on their surface, which interacts with CD47 on tumor cells to inhibit their phagocytic activity. In this study, we developed lipid nanoparticles modified with an anti-SIRPα antibody (aSIRPα) for the targeted delivery of microRNA-155 (miR155@aSIRPα-LNP) to TAMs, aiming to enhance their anti-tumoral phenotypes within the tumor microenvironment. The aSIRPα modification not only facilitated nanoparticle uptake by TAMs rather than B16F10 cells, but also blocked the anti-phagocytosis signal by disrupting the interaction between SIRPα and CD47 on cancer cells. This dual functionality enhanced the expression of anti-tumoral phenotype markers in TAMs and activated macrophage-mediated phagocytosis of tumor cells. In a melanoma model, intratumoral administration of miR155@aSIRPα-LNP to B16F10 tumor-bearing mice reprogrammed TAMs toward anti-tumoral phenotypes. The anti-tumoral cytokines released by these TAMs remodeled the immunosuppressive tumor microenvironment, increasing cytotoxic T cell infiltration and reducing the regulatory T cell population, inhibiting tumor progression. This approach indicates the potential of miRNA-based therapies to overcome the limitations of current immunotherapies in treating cold solid tumors. Overall, the results suggest that delivering miR155 to TAMs by targeting SIRPα is a promising strategy for modulating the immunosuppressive tumor microenvironment in cancer immunotherapy.

求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Controlled Release
医学-化学综合
CiteScore
18.50
自引率
5.60%
发文量
700
审稿时长
39 days
期刊介绍:
The Journal of Controlled Release (JCR) proudly serves as the Official Journal of the Controlled Release Society and the Japan Society of Drug Delivery System.
Dedicated to the broad field of delivery science and technology, JCR publishes high-quality research articles covering drug delivery systems and all facets of formulations. This includes the physicochemical and biological properties of drugs, design and characterization of dosage forms, release mechanisms, in vivo testing, and formulation research and development across pharmaceutical, diagnostic, agricultural, environmental, cosmetic, and food industries.
Priority is given to manuscripts that contribute to the fundamental understanding of principles or demonstrate the advantages of novel technologies in terms of safety and efficacy over current clinical standards. JCR strives to be a leading platform for advancements in delivery science and technology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


