Nickel-catalyzed CN bond formation of diarylamine between nitroarenes and aryl Grignard reagents
IF 6.5
1区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
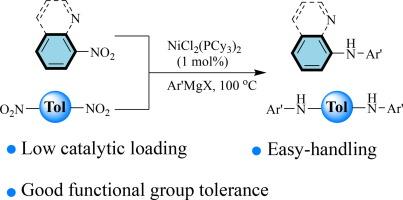
In recent years, transition-metal-catalyzed C![]() N bond formation from nitroarenes and aryl Grignard reagents has emerged as a promising and highly efficient method. Despite this, dinitro aromatic compounds have received limited attention in this context. Herein, we disclose a novel approach for synthesizing diarylamines via Ni-catalyzed cross-coupling of Grignard reagents with both mono- and di-nitro aromatic compounds. This method demonstrates remarkable tolerance towards a wide range of functional groups, including COOEt, F, Cl, CN, CF3, OCF3, SCH3, and pyridyl groups, allowing for the synthesis of various aminated arenes in moderate to good yields. This C
N bond formation from nitroarenes and aryl Grignard reagents has emerged as a promising and highly efficient method. Despite this, dinitro aromatic compounds have received limited attention in this context. Herein, we disclose a novel approach for synthesizing diarylamines via Ni-catalyzed cross-coupling of Grignard reagents with both mono- and di-nitro aromatic compounds. This method demonstrates remarkable tolerance towards a wide range of functional groups, including COOEt, F, Cl, CN, CF3, OCF3, SCH3, and pyridyl groups, allowing for the synthesis of various aminated arenes in moderate to good yields. This C![]() N bond formation method offers a general and step-economical pathway to diaryl and polyaryl amines, paving the way for new synthetic possibilities.
N bond formation method offers a general and step-economical pathway to diaryl and polyaryl amines, paving the way for new synthetic possibilities.

求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Catalysis
工程技术-工程:化工
CiteScore
12.30
自引率
5.50%
发文量
447
审稿时长
31 days
期刊介绍:
The Journal of Catalysis publishes scholarly articles on both heterogeneous and homogeneous catalysis, covering a wide range of chemical transformations. These include various types of catalysis, such as those mediated by photons, plasmons, and electrons. The focus of the studies is to understand the relationship between catalytic function and the underlying chemical properties of surfaces and metal complexes.
The articles in the journal offer innovative concepts and explore the synthesis and kinetics of inorganic solids and homogeneous complexes. Furthermore, they discuss spectroscopic techniques for characterizing catalysts, investigate the interaction of probes and reacting species with catalysts, and employ theoretical methods.
The research presented in the journal should have direct relevance to the field of catalytic processes, addressing either fundamental aspects or applications of catalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


