EHBP1 suppresses liver fibrosis in metabolic dysfunction-associated steatohepatitis
IF 27.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
Excess cholesterol accumulation contributes to fibrogenesis in metabolic dysfunction-associated steatohepatitis (MASH), but how hepatic cholesterol metabolism becomes dysregulated in MASH is not completely understood. We show that human fibrotic MASH livers have decreased EH-domain-binding protein 1 (EHBP1), a genome-wide association study (GWAS) locus associated with low-density lipoprotein (LDL) cholesterol, and that EHBP1 loss- and gain-of-function increase and decrease MASH fibrosis in mice, respectively. Mechanistic studies reveal that EHBP1 promotes sortilin-mediated PCSK9 secretion, leading to LDL receptor (LDLR) degradation, decreased LDL uptake, and reduced TAZ, a fibrogenic effector. At a cellular level, EHBP1 deficiency affects the intracellular localization of retromer, a protein complex required for sortilin stabilization. Our therapeutic approach to stabilizing retromer is effective in mitigating MASH fibrosis. Moreover, we show that the tumor necrosis factor alpha (TNF-α)/peroxisome proliferator-activated receptor alpha (PPARα) pathway suppresses EHBP1 in MASH. These data not only provide mechanistic insights into the role of EHBP1 in cholesterol metabolism and MASH fibrosis but also elucidate an interplay between inflammation and EHBP1-mediated cholesterol metabolism.
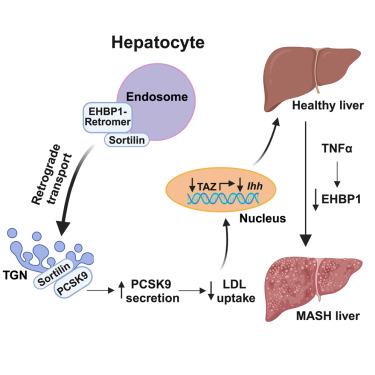
EHBP1抑制代谢功能障碍相关脂肪性肝炎的肝纤维化
过量的胆固醇积累有助于代谢功能障碍相关脂肪性肝炎(MASH)的纤维形成,但在MASH中肝脏胆固醇代谢如何变得失调尚不完全清楚。我们发现,人类纤维化的MASH肝脏中eh结构域结合蛋白1 (EHBP1)减少,EHBP1是一种与低密度脂蛋白(LDL)胆固醇相关的全基因组关联研究(GWAS)位点,EHBP1的功能缺失和功能获得分别增加和减少小鼠的MASH纤维化。机制研究表明,EHBP1促进sortinin介导的PCSK9分泌,导致LDL受体(LDLR)降解,LDL摄取减少,TAZ(一种纤维化效应物)减少。在细胞水平上,EHBP1缺陷影响逆转录物的细胞内定位,逆转录物是sortilin稳定所需的一种蛋白质复合物。我们稳定逆转录酶的治疗方法在缓解MASH纤维化方面是有效的。此外,我们发现肿瘤坏死因子α (TNF-α)/过氧化物酶体增殖物激活受体α (PPARα)通路抑制MASH中的EHBP1。这些数据不仅为EHBP1在胆固醇代谢和MASH纤维化中的作用提供了机制见解,而且阐明了炎症与EHBP1介导的胆固醇代谢之间的相互作用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell metabolism
生物-内分泌学与代谢
CiteScore
48.60
自引率
1.40%
发文量
173
审稿时长
2.5 months
期刊介绍:
Cell Metabolism is a top research journal established in 2005 that focuses on publishing original and impactful papers in the field of metabolic research.It covers a wide range of topics including diabetes, obesity, cardiovascular biology, aging and stress responses, circadian biology, and many others.
Cell Metabolism aims to contribute to the advancement of metabolic research by providing a platform for the publication and dissemination of high-quality research and thought-provoking articles.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


