Astrocyte heterogeneity reveals region-specific astrogenesis in the white matter
IF 21.2
1区 医学
Q1 NEUROSCIENCES
引用次数: 0
Abstract
Astrocyte heterogeneity has been well explored, but our understanding of white matter (WM) astrocytes and their distinctions from gray matter (GM) astrocytes remains limited. Here, we compared astrocytes from cortical GM and WM/corpus callosum (WM/CC) using single-cell RNA sequencing and spatial transcriptomics of the murine forebrain. The comparison revealed similarities but also significant differences between WM and GM astrocytes, including cytoskeletal and metabolic hallmarks specific to WM astrocytes with molecular properties also shared with human WM astrocytes. When we compared murine astrocytes from two different WM regions, the cortex and cerebellum, we found that they exhibited distinct, region-specific molecular properties, with the cerebellum lacking, for example, a specific cluster of WM astrocytes expressing progenitor and proliferation genes. Functional experiments confirmed astrocyte proliferation in the WM/CC, but not in the cerebellar WM, suggesting that the WM/CC may be a source of continued astrogenesis. White matter (WM) astrocytes differ significantly from gray matter astrocytes, with WM astrocytes in the forebrain exhibiting unique proliferation capacity, which is absent in cerebellar WM, suggesting region-specific astrocyte generation.

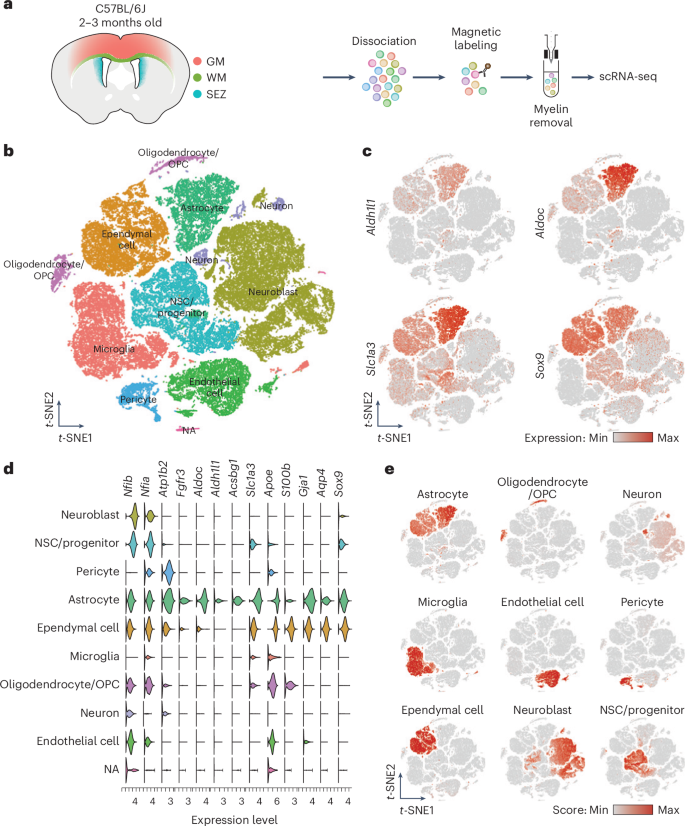
星形胶质细胞的异质性已经得到了很好的探讨,但我们对白质(WM)星形胶质细胞及其与灰质(GM)星形胶质细胞的区别的了解仍然有限。在这里,我们利用小鼠前脑的单细胞 RNA 测序和空间转录组学比较了皮质 GM 和 WM/胼胝体(WM/CC)的星形胶质细胞。比较结果表明,WM 和 GM 星形胶质细胞之间既有相似之处,也有显著差异,包括 WM 星形胶质细胞特有的细胞骨架和代谢特征,以及与人类 WM 星形胶质细胞相同的分子特性。当我们比较来自大脑皮层和小脑这两个不同 WM 区域的小鼠星形胶质细胞时,我们发现它们表现出不同的、区域特异性的分子特性,例如,小脑缺乏表达祖细胞和增殖基因的特定 WM 星形胶质细胞群。功能实验证实了WM/CC中的星形胶质细胞增殖,而小脑WM中的星形胶质细胞没有增殖,这表明WM/CC可能是星形胶质细胞持续生成的来源。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature neuroscience
医学-神经科学
CiteScore
38.60
自引率
1.20%
发文量
212
审稿时长
1 months
期刊介绍:
Nature Neuroscience, a multidisciplinary journal, publishes papers of the utmost quality and significance across all realms of neuroscience. The editors welcome contributions spanning molecular, cellular, systems, and cognitive neuroscience, along with psychophysics, computational modeling, and nervous system disorders. While no area is off-limits, studies offering fundamental insights into nervous system function receive priority.
The journal offers high visibility to both readers and authors, fostering interdisciplinary communication and accessibility to a broad audience. It maintains high standards of copy editing and production, rigorous peer review, rapid publication, and operates independently from academic societies and other vested interests.
In addition to primary research, Nature Neuroscience features news and views, reviews, editorials, commentaries, perspectives, book reviews, and correspondence, aiming to serve as the voice of the global neuroscience community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


