Systemic HER3 ligand-mimicking nanobioparticles enter the brain and reduce intracranial tumour growth
IF 34.9
1区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
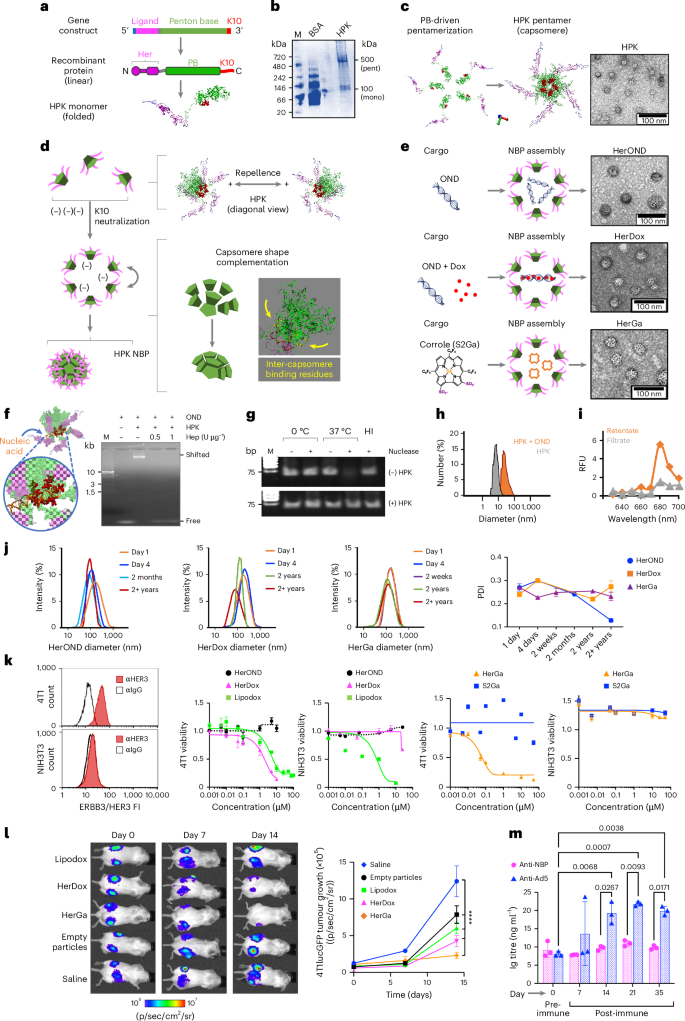
Crossing the blood–brain barrier (BBB) and reaching intracranial tumours is a clinical challenge for current targeted interventions including antibody-based therapies, contributing to poor patient outcomes. Increased cell surface density of human epidermal growth factor receptor 3 (HER3) is associated with a growing number of metastatic tumour types and is observed on tumour cells that acquire resistance to a growing number of clinical targeted therapies. Here we describe the evaluation of HER3-homing nanobiological particles (nanobioparticles (NBPs)) on such tumours in preclinical models and our discovery that systemic NBPs could be found in the brain even in the absence of such tumours. Our subsequent studies described here show that HER3 is prominently associated with both mouse and human brain endothelium and with extravasation of systemic NBPs in mice and in human-derived BBB chips in contrast to non-targeted agents. In mice, systemically delivered NBPs carrying tumoricidal agents reduced the growth of intracranial triple-negative breast cancer cells, which also express HER3, with improved therapeutic profile compared to current therapies and compared to agents using traditional BBB transport routes. As HER3 associates with a growing number of metastatic tumours, the NBPs described here may offer targeted efficacy especially when such tumours localize to the brain. Delivering therapeutics to the brain is challenging because of the hard-to-cross blood–brain barrier. Here, the authors show that HER3, which is expressed on the surface of many metastatic tumours, is associated with the brain endothelium and can drive accumulation of HER3-targeted nanoparticles within the brain, for therapy against HER3-positive tumours.

系统模拟HER3配体的纳米生物颗粒进入大脑并减少颅内肿瘤的生长
通过血脑屏障(BBB)到达颅内肿瘤是目前靶向干预的临床挑战,包括基于抗体的治疗,导致患者预后不佳。人表皮生长因子受体3 (HER3)细胞表面密度的增加与越来越多的转移性肿瘤类型相关,并且在对越来越多的临床靶向治疗产生耐药性的肿瘤细胞上观察到。在这里,我们描述了her3定位纳米生物颗粒(纳米生物颗粒(nbp))在临床前模型中对此类肿瘤的评估,以及我们的发现,即使在没有此类肿瘤的情况下,也可以在大脑中发现系统性的nbp。我们在此描述的后续研究表明,与非靶向药物相比,HER3与小鼠和人的脑内皮以及小鼠和人源性血脑屏障芯片的全身NBPs外渗都有显著相关性。在小鼠中,与目前的治疗方法和使用传统血脑屏障运输途径的药物相比,系统递送携带杀瘤剂的NBPs减少了颅内三阴性乳腺癌细胞的生长,这些细胞也表达HER3,具有更好的治疗效果。由于HER3与越来越多的转移性肿瘤相关,本文描述的nbp可能提供靶向疗效,特别是当此类肿瘤局限于大脑时。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature nanotechnology
工程技术-材料科学:综合
CiteScore
59.70
自引率
0.80%
发文量
196
审稿时长
4-8 weeks
期刊介绍:
Nature Nanotechnology is a prestigious journal that publishes high-quality papers in various areas of nanoscience and nanotechnology. The journal focuses on the design, characterization, and production of structures, devices, and systems that manipulate and control materials at atomic, molecular, and macromolecular scales. It encompasses both bottom-up and top-down approaches, as well as their combinations.
Furthermore, Nature Nanotechnology fosters the exchange of ideas among researchers from diverse disciplines such as chemistry, physics, material science, biomedical research, engineering, and more. It promotes collaboration at the forefront of this multidisciplinary field. The journal covers a wide range of topics, from fundamental research in physics, chemistry, and biology, including computational work and simulations, to the development of innovative devices and technologies for various industrial sectors such as information technology, medicine, manufacturing, high-performance materials, energy, and environmental technologies. It includes coverage of organic, inorganic, and hybrid materials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


