A phase III randomised study of first-line NUC-1031/cisplatin vs. gemcitabine/cisplatin in advanced biliary tract cancer
IF 26.8
1区 医学
Q1 GASTROENTEROLOGY & HEPATOLOGY
引用次数: 0
Abstract
Background and Aims
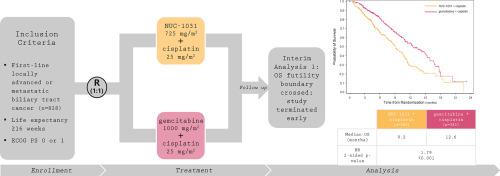
The previous first-line standard of care for advanced biliary tract cancer (aBTC), gemcitabine/cisplatin, had modest efficacy. NUC-1031 is a phosphoramidate modification of gemcitabine. We report final analyses of the NuTide:121 study, designed to compare the efficacy of NUC-1031/cisplatin to gemcitabine/cisplatin in aBTC.Methods
In this open-label, multicenter study, adult patients with treatment-naïve aBTC were randomized (1:1) to NUC-1031/cisplatin (n=388) or gemcitabine/cisplatin (n=385) on Days 1 and 8 of 21-day cycles until disease progression or intolerance. Primary endpoints were overall survival (OS) and objective response rate (ORR; blinded independent central review). Three interim analyses (IA) and a final analysis were planned.Results
Baseline characteristics were balanced; median age 65 years, 53% male, primary tumors: intra-hepatic cholangiocarcinoma (CCA) (54%), extra-hepatic CCA (21%), gallbladder (21%) and ampullary cancer (5%). Enrollment stopped at IA1 as the OS futility boundary was crossed. At final data cut-off, median OS for NUC-1031/cisplatin vs gemcitabine/cisplatin was 9.2 months (95%CI: 8.3–10.4) vs 12.6 months (95%CI: 11.0–15.1) (HR 1.79) and median PFS was 4.9 months (95%CI: 4.4–6.0) vs 6.4 months (95%CI: 6.1–7.4) (HR 1.45). ORR was higher for NUC-1031/cisplatin (18.7% vs 12.4%; OR: 1.59; p=0.049). The adverse event profile was similar between arms, except for hepatobiliary disorders (25% vs 11%; higher with NUC-1031/cisplatin) and hematological events (48% vs 65%; higher with gemcitabine/cisplatin). More patients met criteria for potential drug-induced liver injury (27% vs 7%) and Hy’s law (1.6% vs 0.5%) with NUC-1031/cisplatin. Treatment exposure was lower for NUC-1031/cisplatin, likely due to early discontinuation for AEs (30% vs 18%).Conclusions
NuTide:121 was terminated early due to futility. NUC-1031/cisplatin did not set a new standard in first-line aBTC.Trial Registration
ClinicalTrials.gov Identifier: NCT04163900Impact and Implications
Advanced biliary tract cancers remain in need of substantial therapeutic improvement. Although clinical practice guidelines identified gemcitabine plus cisplatin as the standard of care on the basis of clinical studies, the modest efficacy observed with this regimen highlighted the urgent need for more effective therapies. NuTide:121, one of the largest randomized interventional studies conducted in the first-line aBTC population to date, compared the combination of cisplatin with NUC-1031, a phosphoramidate form of gemcitabine, with the standard of care regimen. Despite a higher response rate in the NUC-1031/cisplatin arm, the study was terminated early based on a futility assessment of OS. Early toxicity and in particular liver injury likely contributed to the regimen's failure. This study emphasized some important challenges in study design and further confirmed the difficulties of advancing treatment options in this vulnerable patient population.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Hepatology
医学-胃肠肝病学
CiteScore
46.10
自引率
4.30%
发文量
2325
审稿时长
30 days
期刊介绍:
The Journal of Hepatology is the official publication of the European Association for the Study of the Liver (EASL). It is dedicated to presenting clinical and basic research in the field of hepatology through original papers, reviews, case reports, and letters to the Editor. The Journal is published in English and may consider supplements that pass an editorial review.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


