Photo-induced aerobic deborohydroxylation of organoboron compounds in 1,3-dioxolane at room temperature
IF 6.5
1区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
We report herein a simple and efficient light-induced aerobic oxidation of organoboron compounds to obtain phenols in an air atmosphere at room temperature, without the use of any photocatalyst, oxidant or other reagent. This protocol utilizes inexpensive and commercially available 1,3-dioxolane as both the solvent and electron donor to activate the molecular oxygen in the air under 365 nm light, enabling the aerobic oxidation to occur under mild reaction conditions. A wide range of sensitive functional groups are well tolerated, resulting in the corresponding phenols in moderate to good yields.

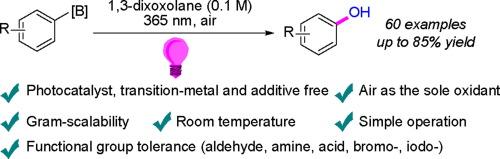
室温下有机硼化合物在 1,3-二氧戊环中的光诱导有氧脱硼羟基反应
我们在此报告一种简单高效的光诱导有机硼化合物有氧氧化方法,该方法无需使用任何光催化剂、氧化剂或其他试剂,即可在室温空气环境中获得苯酚。该方案利用市场上廉价的 1,3-二氧戊环作为溶剂和电子供体,在 365 纳米波长的光照下激活空气中的分子氧,从而在温和的反应条件下实现有氧氧化。该方法对多种敏感官能团都有很好的耐受性,并能以中等到良好的收率制备出相应的苯酚。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Catalysis
工程技术-工程:化工
CiteScore
12.30
自引率
5.50%
发文量
447
审稿时长
31 days
期刊介绍:
The Journal of Catalysis publishes scholarly articles on both heterogeneous and homogeneous catalysis, covering a wide range of chemical transformations. These include various types of catalysis, such as those mediated by photons, plasmons, and electrons. The focus of the studies is to understand the relationship between catalytic function and the underlying chemical properties of surfaces and metal complexes.
The articles in the journal offer innovative concepts and explore the synthesis and kinetics of inorganic solids and homogeneous complexes. Furthermore, they discuss spectroscopic techniques for characterizing catalysts, investigate the interaction of probes and reacting species with catalysts, and employ theoretical methods.
The research presented in the journal should have direct relevance to the field of catalytic processes, addressing either fundamental aspects or applications of catalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


