Borrowing hydrogen alkylation of indenes with alcohols by manganese pincer complex
IF 6.5
1区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
Herein, we report a highly efficient and selective formal C3-alkylation of indenes with a wide range of primary and secondary alcohols catalyzed by PNP-manganese pincer complex. This transformation proceeds via a borrowing hydrogen strategy, utilizing sustainable and abundant alcohols as alkylating agents. The reaction offers good functional group tolerance and regioselectivity, providing a practical and straightforward approach to the synthesis of alkylated indenes. Preliminary mechanistic investigations indicate that the reaction proceeds via a multi-step pathway involving the dehydrogenation of the alcohol to a carbonyl intermediate, followed by cross-condensation, hydrogenation, and isomerization steps to afford the final product.

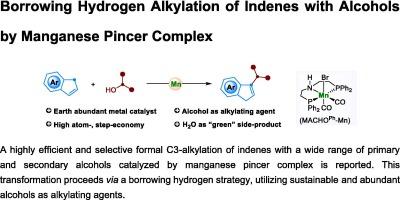
在此,我们报告了在 PNP-锰钳形复合物催化下,茚与多种伯醇和仲醇发生的高效、选择性的 C3-正式烷基化反应。这种转化通过借氢策略进行,利用可持续的丰富醇类作为烷化剂。该反应具有良好的官能团耐受性和区域选择性,为合成烷基化茚提供了一种实用而直接的方法。初步的机理研究表明,该反应通过多步途径进行,包括醇脱氢生成羰基中间体,然后经过缩合、氢化和异构化步骤得到最终产物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Catalysis
工程技术-工程:化工
CiteScore
12.30
自引率
5.50%
发文量
447
审稿时长
31 days
期刊介绍:
The Journal of Catalysis publishes scholarly articles on both heterogeneous and homogeneous catalysis, covering a wide range of chemical transformations. These include various types of catalysis, such as those mediated by photons, plasmons, and electrons. The focus of the studies is to understand the relationship between catalytic function and the underlying chemical properties of surfaces and metal complexes.
The articles in the journal offer innovative concepts and explore the synthesis and kinetics of inorganic solids and homogeneous complexes. Furthermore, they discuss spectroscopic techniques for characterizing catalysts, investigate the interaction of probes and reacting species with catalysts, and employ theoretical methods.
The research presented in the journal should have direct relevance to the field of catalytic processes, addressing either fundamental aspects or applications of catalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


