para-Selective C–H alkylation of aroyl chlorides through organic photoredox-catalyzed radical tele-substitution
IF 19.1
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
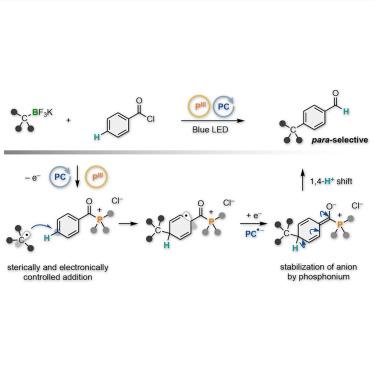
The site-selective C(sp2)–H alkylation of arenes has provided ubiquitous fragments found in pharmaceutical drugs and agrochemicals. Although ortho- and meta-selective alkylation reactions have been achieved well, para-selective examples are scarce. The currently available methods have been designed on the unique behaviors and characteristics of metals to achieve high para-selectivity. Herein, we demonstrate the para-selective C(sp2)–H alkylation of aroyl chlorides with alkyltrifluoroborates and trialkylphosphine through an organic photoredox-catalyzed radical tele-substitution. The reaction proceeds through the para-selective addition of alkyl radicals to in-situ-generated aroyl trialkylphosphonium intermediates followed by a hydrogen shift. Notably, the para-alkylation is associated with the conversion of the carbonyl trialkylphosphonium group to the formyl group, producing para-alkylated benzaldehyde derivatives. This metal-free site-selective C(sp2)–C(sp3) bond formation allows for streamlined synthesis of siponimod and SR-31747.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chem
Environmental Science-Environmental Chemistry
CiteScore
32.40
自引率
1.30%
发文量
281
期刊介绍:
Chem, affiliated with Cell as its sister journal, serves as a platform for groundbreaking research and illustrates how fundamental inquiries in chemistry and its related fields can contribute to addressing future global challenges. It was established in 2016, and is currently edited by Robert Eagling.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


