Unveiling the Molecular Legacy of Transient Insulin Resistance: Implications for Hepatic Metabolic Adaptability
IF 26.8
1区 医学
Q1 GASTROENTEROLOGY & HEPATOLOGY
引用次数: 0
Abstract
Background
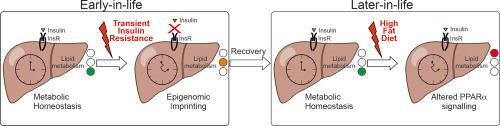
Metabolic flexibility (MetF) is an organism's ability to adjust to changing metabolic supplies and energy demands. Insulin plays a central role in coordinating MetF through molecular mechanisms such as signaling pathways, transcriptional responses, and circadian regulation. Insulin resistance (IR) can impair MetF, contributing to type 2 diabetes and obesity, often stemming from continuous challenges such as sedentary lifestyles, poor diets, and circadian disruptions. Transient IR episodes, like gestational diabetes or stress-induced hyperglycemia, also heighten the risk of later diabetes development. Yet, the molecular processes post-transient IR remain poorly understood despite their health significance.Aims and Methods
Our aims were to characterize the hepatic response to a high fat diet challenge in mice previously exposed to a transient IR episode. We integrated transcriptomic, epigenomic, lipidomic, and molecular clock assessments to provide a molecular basis for the observed dysregulations.Results
Our study shows that temporarily blocking the insulin receptor in young mice leads to later-life liver issues hindering PPARα-mediated adaptation to a high-fat diet. This is linked to decreased histone active marks at PPARα sites and reduced endogenous PPARα ligands. Transient insulin receptor blockade also altered the liver's molecular clock, particularly affecting PPARα transcriptional responsiveness.Conclusions
Seemingly reversible and unnoticed metabolic challenges in early adulthood may predispose the liver to exacerbated metabolic dysfunctions when confronted with chronic challenges later in life.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Hepatology
医学-胃肠肝病学
CiteScore
46.10
自引率
4.30%
发文量
2325
审稿时长
30 days
期刊介绍:
The Journal of Hepatology is the official publication of the European Association for the Study of the Liver (EASL). It is dedicated to presenting clinical and basic research in the field of hepatology through original papers, reviews, case reports, and letters to the Editor. The Journal is published in English and may consider supplements that pass an editorial review.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


