STAT3-controlled CHI3L1/SPP1 positive feedback loop demonstrates the spatial heterogeneity and immune characteristics of glioblastoma
IF 10.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
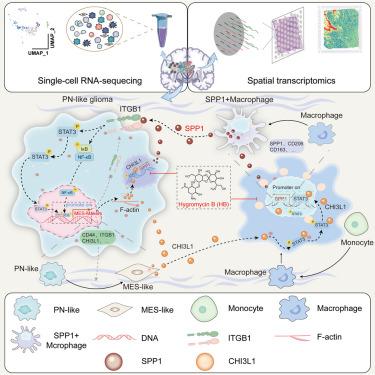
Proneural-mesenchymal transition (PMT) is a phenotypic alteration and contributes to the malignant progression of glioblastoma (GBM). Macrophages, as a main infiltrating component of the tumor immune microenvironment (TIM), control the biological processes of PMT; however, the mechanisms driving this process remain largely unknown. Here, the overall landscape of tumor and nontumor cells was described by scMulti-omics technology. Then, we demonstrated that chitinase-3-like protein 1 (CHI3L1) played a critical role in maintaining mesenchymal (MES) status and reprogramming macrophage phenotype using C57BL/6 and NSG mice models derived from PN20 cells. Mechanistically, osteopontin (OPN)/ITGB1 maintained the activation of nuclear factor κB (NF-κB) and signal transducer and activator of transcription 3 (STAT3) pathways by establishing a positive feedback loop with the CHI3L1-STAT3 axis, resulting in PMT. CHI3L1 enhanced the phosphorylation, nuclear localization, and transcriptional activity of STAT3 via directly binding its coiled-coil domain (CCD). Importantly, we screened and validated that hygromycin B (HB), an inhibitor of the STAT3-CCD domain, disrupted the CHI3L1-STAT3 interaction, thereby reducing the tumor burden in vitro and in vivo.
求助全文
约1分钟内获得全文
求助全文
来源期刊
Developmental cell
生物-发育生物学
CiteScore
18.90
自引率
1.70%
发文量
203
审稿时长
3-6 weeks
期刊介绍:
Developmental Cell, established in 2001, is a comprehensive journal that explores a wide range of topics in cell and developmental biology. Our publication encompasses work across various disciplines within biology, with a particular emphasis on investigating the intersections between cell biology, developmental biology, and other related fields. Our primary objective is to present research conducted through a cell biological perspective, addressing the essential mechanisms governing cell function, cellular interactions, and responses to the environment. Moreover, we focus on understanding the collective behavior of cells, culminating in the formation of tissues, organs, and whole organisms, while also investigating the consequences of any malfunctions in these intricate processes.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


