De novo designed Hsp70 activator dissolves intracellular condensates
IF 7.2
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Protein quality control (PQC) is carried out in part by the chaperone Hsp70 in concert with adapters of the J-domain protein (JDP) family. The JDPs, also called Hsp40s, are thought to recruit Hsp70 into complexes with specific client proteins. However, the molecular principles regulating this process are not well understood. We describe the de novo design of Hsp70 binding proteins that either inhibit or stimulate Hsp70 ATPase activity. An ATPase stimulating design promoted the refolding of denatured luciferase in vitro, similar to native JDPs. Targeting of this design to intracellular condensates resulted in their nearly complete dissolution and revealed roles as cell growth promoting signaling hubs. The designs inform our understanding of chaperone structure-function relationships and provide a general and modular way to target PQC systems to regulate condensates and other cellular targets.
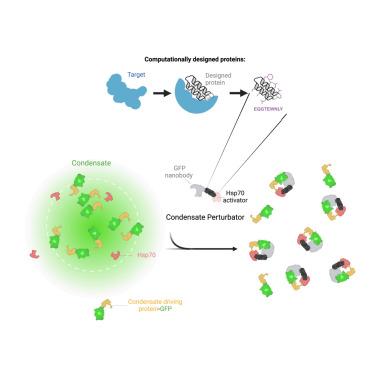

全新设计的Hsp70激活剂可溶解细胞内凝析物
蛋白质量控制(PQC)部分由伴侣蛋白Hsp70与j结构域蛋白(JDP)家族的适配器一起进行。jdp,也被称为hsp40,被认为将Hsp70招募到与特定客户蛋白的复合物中。然而,调控这一过程的分子原理尚不清楚。我们描述了抑制或刺激Hsp70 atp酶活性的Hsp70结合蛋白的重新设计。atp酶刺激设计在体外促进变性荧光素酶的再折叠,类似于天然jdp。这种设计的目标是细胞内凝聚物,导致它们几乎完全溶解,并揭示了作为细胞生长促进信号中枢的作用。这些设计使我们了解了伴侣分子的结构功能关系,并提供了一种通用的模块化方法来靶向PQC系统来调节凝析物和其他细胞靶标。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cell Chemical Biology
Biochemistry, Genetics and Molecular Biology-Molecular Medicine
CiteScore
14.70
自引率
2.30%
发文量
143
期刊介绍:
Cell Chemical Biology, a Cell Press journal established in 1994 as Chemistry & Biology, focuses on publishing crucial advances in chemical biology research with broad appeal to our diverse community, spanning basic scientists to clinicians. Pioneering investigations at the chemistry-biology interface, the journal fosters collaboration between these disciplines. We encourage submissions providing significant conceptual advancements of broad interest across chemical, biological, clinical, and related fields. Particularly sought are articles utilizing chemical tools to perturb, visualize, and measure biological systems, offering unique insights into molecular mechanisms, disease biology, and therapeutics.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


