Photooxidation of Dipyrrinones: Reaction with Singlet Oxygen and Characterization of Reaction Intermediates
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
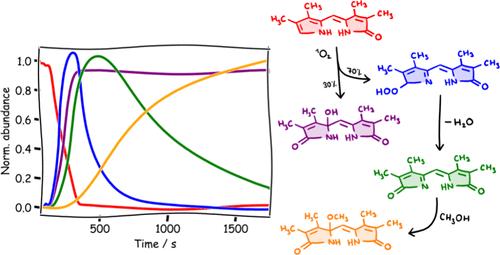
Bilirubin (BR) is a water-insoluble product of heme catabolism in mammals. Elevated blood concentrations of BR, especially in the neonatal period, are treated with blue-green light phototherapy. The major mechanism of BR elimination during phototherapy is photoisomerization, while a minor, less studied mechanism of degradation is oxidation. In this work, we studied the oxidation of the bilirubin model tetramethyl-dipyrrinone (Z-13) by singlet oxygen in methanol using UV–vis and ESI-MS spectroscopy, resulting in propentdyopents as the main oxidation products. We also identified two additional intermediates that were formed during the reaction (hydroperoxide 21a and imine 17). The structure of the hydroperoxide was confirmed by helium-tagging IR spectroscopy. Such reaction intermediates formed during the oxidation of BR or bilirubin models have not been described so far. We believe that this work can be used as a first step in studying the complex oxidation mechanism of BR during phototherapy.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


