Understanding Protein Adsorption on Carbon Nanotube Inner and Outer Surfaces by Molecular Dynamics Simulations
IF 3.7
2区 化学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
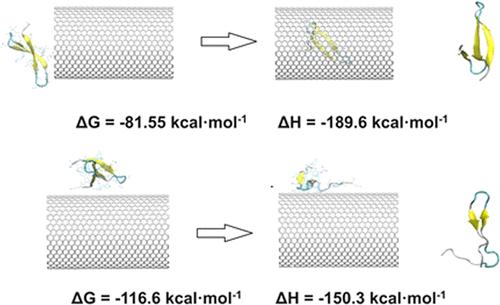
Biomolecules, such as proteins, can form complexes with carbon nanotubes (CNTs), which have numerous applications in nanobiotechnology. Proteins can be adsorbed onto either the inner walls or outer surfaces of CNTs via van der Waals interactions; however, the differences between these two processes remain poorly understood. In this work, we performed classical all-atom molecular dynamics simulations with explicit solvents to investigate the interaction between a model protein, the Yap65 WW domain, and (22,22) CNTs and larger. The Yap65 WW domain comprises three β-sheet segments and contains three key aromatic residues: TRP17, TYR28, and TRP39. Our findings reveal distinct interaction mechanisms for the inner and outer surfaces of large CNTs. The protein’s interaction with the inner surface is governed by the interplay between surface curvature and adsorption orientation. In the confined space of the CNT channel, variations in tube curvature and adsorption orientation give rise to specific binding modes, resulting in varying degrees of protein conformational change. In contrast, on the outer surface of large CNTs, where space is less restricted, the adsorption orientation plays a more dominant role. Specifically, the orientation in which more aromatic residues directly interact with the surface suffer from the greater structural loss, regardless of the tube curvature. Finally, protein-CNT binding free energies were calculated using the Poisson–Boltzmann surface area (MM-PBSA) method and steered molecular dynamics simulations based on Jarzynski equality, demonstrating that protein desorption from CNTs is highly dependent on binding configurations. This study reveals the influence of confined space on protein adsorption and the critical role of CNT curvature in modulating β-sheet stability.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Langmuir
化学-材料科学:综合
CiteScore
6.50
自引率
10.30%
发文量
1464
审稿时长
2.1 months
期刊介绍:
Langmuir is an interdisciplinary journal publishing articles in the following subject categories:
Colloids: surfactants and self-assembly, dispersions, emulsions, foams
Interfaces: adsorption, reactions, films, forces
Biological Interfaces: biocolloids, biomolecular and biomimetic materials
Materials: nano- and mesostructured materials, polymers, gels, liquid crystals
Electrochemistry: interfacial charge transfer, charge transport, electrocatalysis, electrokinetic phenomena, bioelectrochemistry
Devices and Applications: sensors, fluidics, patterning, catalysis, photonic crystals
However, when high-impact, original work is submitted that does not fit within the above categories, decisions to accept or decline such papers will be based on one criteria: What Would Irving Do?
Langmuir ranks #2 in citations out of 136 journals in the category of Physical Chemistry with 113,157 total citations. The journal received an Impact Factor of 4.384*.
This journal is also indexed in the categories of Materials Science (ranked #1) and Multidisciplinary Chemistry (ranked #5).
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


