Synthesis and Evaluation of Novel Ebselen Derivatives as Urease Inhibitors for Combating Helicobacter pylori Infections
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
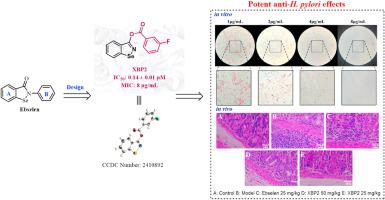
The development of novel inhibitors targeting urease that affects the colonization of Helicobacter pylori (H. pylori) is a promising strategy to address the growing challenge of antibiotic resistance. In this study, a novel urease covalent inhibitor, XBP2 (IC50 = 0.14 ± 0.01 μM and MIC = 8 μg/mL), was identified through structure-based design using Ebselen as a template. XBP2 binds to a newly identified site, forming stable dipole interaction with residues ASP223 and HIS322, which enhances both stability and inhibitory activity against urease. It exhibits potent in vitro antibacterial activity, reducing cell apoptosis rates and significantly decreasing the fluorescence intensity of ROS and γH2AX in GES-1 cells infected with H. pylori. In the mouse gastritis model infected with H. pylori, XBP2 exhibits significant gastric mucosal protective effects. High-dose XBP2 (50 mg/kg) effectively prevented pathological changes such as bleeding and hyperplasia. Furthermore, acute toxicity test revealed that XBP2 does not display any detectable toxicity. These findings not only expand the structural diversity of Ebselen derivatives but also provide valuable insights for developing targeted therapies for eradicating H. pylori.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


