Shigella infection is facilitated by interaction of human enteric α-defensin 5 with colonic epithelial receptor P2Y11
IF 20.5
1区 生物学
Q1 MICROBIOLOGY
引用次数: 0
Abstract
Human enteric α-defensin 5 (HD5) is an immune system peptide that acts as an important antimicrobial factor but is also known to promote pathogen infections by enhancing adhesion of the pathogens. The mechanistic basis of these conflicting functions is unknown. Here we show that HD5 induces abundant filopodial extensions in epithelial cells that capture Shigella, a major human enteroinvasive pathogen that is able to exploit these filopodia for invasion, revealing a mechanism for HD5-augmented bacterial invasion. Using multi-omics screening and in vitro, organoid, dynamic gut-on-chip and in vivo models, we identify the HD5 receptor as P2Y11, a purinergic receptor distributed apically on the luminal surface of the human colonic epithelium. Inhibitor screening identified cAMP-PKA signalling as the main pathway mediating the cytoskeleton-regulating activity of HD5. In illuminating this mechanism of Shigella invasion, our findings raise the possibility of alternative intervention strategies against HD5-augmented infections. HD5 induces filopodial extensions in epithelial cells that the pathogen Shigella exploits to facilitate invasion and infection.

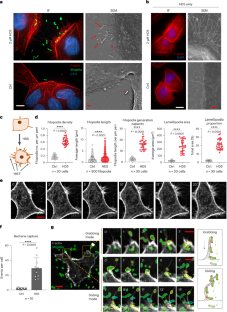
人肠道α-防御素5与结肠上皮受体P2Y11的相互作用促进了志贺氏菌感染
人肠道α-防御素5 (HD5)是一种免疫系统肽,作为一种重要的抗菌因子,但也已知通过增强病原体的粘附来促进病原体感染。这些相互冲突的功能的机制基础是未知的。在这里,我们发现HD5在上皮细胞中诱导了丰富的丝足延伸,这些丝足延伸可以捕获志贺氏菌,志贺氏菌是一种主要的人类肠道侵入性病原体,能够利用这些丝足进行入侵,揭示了HD5增强细菌入侵的机制。通过多组学筛选和体外、类器官、动态肠道芯片和体内模型,我们确定HD5受体为P2Y11,一种分布在人类结肠上皮管腔表面的嘌呤能受体。抑制剂筛选发现cAMP-PKA信号通路是介导HD5细胞骨架调节活性的主要途径。在阐明志贺氏菌入侵的这一机制时,我们的发现提出了针对hd5增强感染的替代干预策略的可能性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Microbiology
Immunology and Microbiology-Microbiology
CiteScore
44.40
自引率
1.10%
发文量
226
期刊介绍:
Nature Microbiology aims to cover a comprehensive range of topics related to microorganisms. This includes:
Evolution: The journal is interested in exploring the evolutionary aspects of microorganisms. This may include research on their genetic diversity, adaptation, and speciation over time.
Physiology and cell biology: Nature Microbiology seeks to understand the functions and characteristics of microorganisms at the cellular and physiological levels. This may involve studying their metabolism, growth patterns, and cellular processes.
Interactions: The journal focuses on the interactions microorganisms have with each other, as well as their interactions with hosts or the environment. This encompasses investigations into microbial communities, symbiotic relationships, and microbial responses to different environments.
Societal significance: Nature Microbiology recognizes the societal impact of microorganisms and welcomes studies that explore their practical applications. This may include research on microbial diseases, biotechnology, or environmental remediation.
In summary, Nature Microbiology is interested in research related to the evolution, physiology and cell biology of microorganisms, their interactions, and their societal relevance.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


