An avoidance segment resolves a lethal nuclear–mitochondrial targeting conflict during ribosome assembly
IF 17.3
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
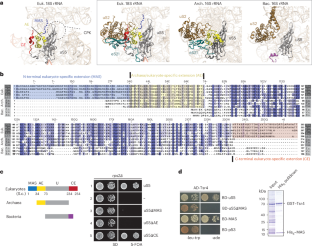
The correct sorting of nascent ribosomal proteins from the cytoplasm to the nucleus or to mitochondria for ribosome production poses a logistical challenge for cellular targeting pathways. Here we report the discovery of a conserved mitochondrial avoidance segment (MAS) within the cytosolic ribosomal protein uS5 that resolves an evolutionary lethal conflict between the nuclear and mitochondrial targeting machinery. MAS removal mistargets uS5 to the mitochondrial matrix and disrupts the assembly of the cytosolic ribosome. The resulting lethality can be rescued by impairing mitochondrial import. We show that MAS triages nuclear targeting by disabling a cryptic mitochondrial targeting activity within uS5 and thereby prevents fatal capture by mitochondria. Our findings identify MAS as an essential acquisition by the primordial eukaryote that reinforced organelle targeting fidelity while developing an endosymbiotic relationship with its mitochondrial progenitor. Oborská-Oplová et al. report a conserved mitochondrial avoidance segment in the cytosolic ribosomal protein uS5 that prevents mistargeting of uS5 to the mitochondrial matrix and ensures cytosolic ribosomal assembly.

求助全文
约1分钟内获得全文
求助全文
来源期刊
Nature Cell Biology
生物-细胞生物学
CiteScore
28.40
自引率
0.90%
发文量
219
审稿时长
3 months
期刊介绍:
Nature Cell Biology, a prestigious journal, upholds a commitment to publishing papers of the highest quality across all areas of cell biology, with a particular focus on elucidating mechanisms underlying fundamental cell biological processes. The journal's broad scope encompasses various areas of interest, including but not limited to:
-Autophagy
-Cancer biology
-Cell adhesion and migration
-Cell cycle and growth
-Cell death
-Chromatin and epigenetics
-Cytoskeletal dynamics
-Developmental biology
-DNA replication and repair
-Mechanisms of human disease
-Mechanobiology
-Membrane traffic and dynamics
-Metabolism
-Nuclear organization and dynamics
-Organelle biology
-Proteolysis and quality control
-RNA biology
-Signal transduction
-Stem cell biology
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


