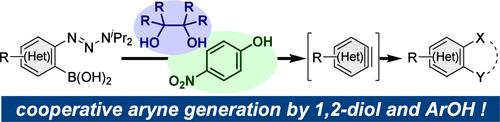
Aryne Generation from o-Triazenylarylboronic Acids Induced by 1,2-Diols and 4-Nitrophenol
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Arynes are important synthetic intermediates that are usually generated under alkaline conditions. We developed a method for generating arynes using two hydroxy compounds as activators. o-Triazenylarylboronic acids generate (hetero)arynes when activated by a combination of ethylene glycol, pinacol, and p-nitrophenol; these arynes then react with a range of arynophiles under slightly acidic conditions that complement the conventional basic conditions with unique chemoselectivities observed even in the presence of excess hydroxy compounds.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


