In Vitro and In Vivo Anticancer Activities of Water-Soluble Ru(II)(η6-p-cymene) Complexes via Activating Apoptosis Central Regulators and Possibilities of New Antitumor Strategies in Triple Negative Breast Cancers
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
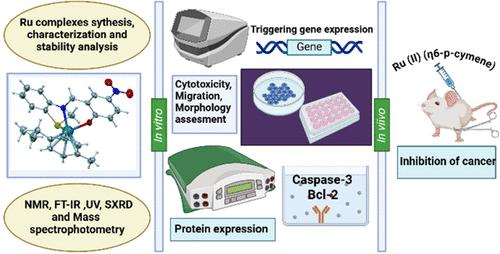
In this study, we synthesized 12 monofunctional tridentate ONS-donor salicylaldimine ligand (L)-based Ru(II) complexes with general formula [(Ru(L)(p-cymene)]+·Cl– (C1-C12), characterized by 1H NMR, 13C NMR, UV, FT-IR spectroscopy, HR-ESI mass spectrometry, and single-crystal X-ray analysis showing ligand’s orientation around the Ru(II) center. All 12 of these 12 complexes were tested for their anticancer activities in multiple cancer cells. The superior antitumor efficacy of C2, C8, and C11 was demonstrated by reduced mitochondrial membrane potential, impaired proliferative capacity, and disrupted redox homeostasis, along with enhanced apoptosis through caspase-3 activation and downregulation of Bcl-2 expression. In the 4T1 breast cancer orthotopic mouse model, assessment of bioluminescence for metastatic spread, tumor burden, histopathological evaluation, immunohistochemistry (IHC), and hematological profiling and tissue Protein expression of caspase-3, cleaved caspase-3, TNF-α, and bcl-2 demonstrated that C8 treatment led to prolonged survival and suppressed tumor progression in triple negative breast cancer.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


