Photooxidation and Cleavage of Ethynylated 9,10-Dimethoxyanthracenes with Acid-Labile Ether Bonds
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
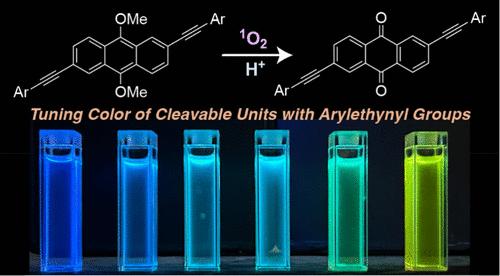
This paper describes a series of 12 9,10-dimethoxyanthracene derivatives functionalized with a range of electronically diverse ethynyl substituents at the 2 and 6 positions, aimed at tuning their optoelectronic properties and reactivity with singlet oxygen (1O2). Optical spectroscopy, cyclic voltammetry, and density functional theory calculations reveal that the ethynyl groups decrease the HOMO–LUMO gaps in these acenes. Notably, bis(dimethylanilineethynyl) substituents increase the wavelength of absorbance onset by over 60 nm compared to 9,10-dimethoxyanthracene (DMA). Furthermore, all 12 molecules react with 1O2 through cycloaddition at the 9 and 10 positions to form endoperoxides. Although the presence of ethynyl groups decreases the reaction rates, they are at least 40% of the rate observed for DMA. Finally, these endoperoxides cleave to form quinones when exposed to protic acid. This behavior, combined with the red-shifting of absorbance spectra, emphasizes their potential in photocleavable materials.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


