Evolution Driven Microscale Combinatorial Chemistry in Intracellular Mimicking Droplets to Engineer Thermostable RNA for Cellular Imaging
IF 13
2区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
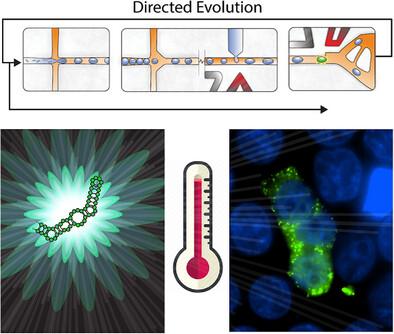
Fluorescent light-up aptamer/fluorogen pairs are powerful tools for tracking RNA in the cell, however limitations in thermostability and fluorescence intensity exist. Current in vitro selection techniques struggle to mimic complex intracellular environments, limiting in vivo biomolecule functionality. Taking inspiration from microenvironment-dependent RNA folding observed in cells and organelle-mimicking droplets, an efficient system is created that uses microscale heated water droplets to simulate intracellular conditions, effectively replicating the intracellular RNA folding landscape. This system is integrated with microfluidic droplet sorting to evolve RNA aptamers. Through this approach, an RNA aptamer is engineered with improved fluorescence activity by exploring the chemical fitness landscape under biomimetic conditions. The enhanced RNA aptamer named eBroccoli has increased fluorescence intensity and thermal stability, both in vitro and in vivo in bacterial and mammalian cells. In mammalian cell culture conditions, a fluorescence improvement of 3.9-times is observed and biological thermal stability up to 45 °C is observed in bacterial systems. eBroccoli enable real-time visualization of nanoscale stress granule formation in mammalian cells during heat shock at 42 °C. By introducing the concept of “biomimetic equivalence” based on RNA folding, the platform offers a simple yet effective strategy to mimic intracellular complexity in evolution-based engineering.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Small
工程技术-材料科学:综合
CiteScore
17.70
自引率
3.80%
发文量
1830
审稿时长
2.1 months
期刊介绍:
Small serves as an exceptional platform for both experimental and theoretical studies in fundamental and applied interdisciplinary research at the nano- and microscale. The journal offers a compelling mix of peer-reviewed Research Articles, Reviews, Perspectives, and Comments.
With a remarkable 2022 Journal Impact Factor of 13.3 (Journal Citation Reports from Clarivate Analytics, 2023), Small remains among the top multidisciplinary journals, covering a wide range of topics at the interface of materials science, chemistry, physics, engineering, medicine, and biology.
Small's readership includes biochemists, biologists, biomedical scientists, chemists, engineers, information technologists, materials scientists, physicists, and theoreticians alike.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


