Nanoscale Cellular Traction Force Quantification: CRISPR-Cas12a Supercharged DNA Tension Sensors in Nanoclustered Ligand Patterns
IF 8.3
2区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
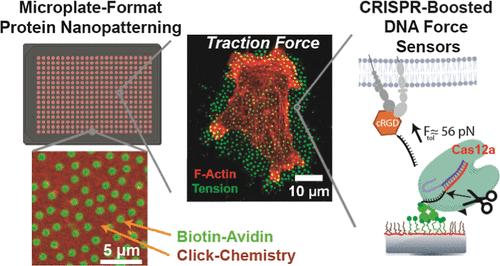
High-throughput measurement of cellular traction forces at the nanoscale remains a significant challenge in mechanobiology, limiting our understanding of how cells interact with their microenvironment. Here, we present a novel technique for fabricating protein nanopatterns in standard multiwell microplate formats (96/384-wells), enabling the high-throughput quantification of cellular forces using DNA tension gauge tethers (TGTs) amplified by CRISPR-Cas12a. Our method employs sparse colloidal lithography to create nanopatterned surfaces with feature sizes ranging from sub 100 to 800 nm on transparent, planar, and fully PEGylated substrates. These surfaces allow for the orthogonal immobilization of two different proteins or biomolecules using click-chemistry, providing precise spatial control over cellular signaling cues. We demonstrate the robustness and versatility of this platform through imaging techniques, including total internal reflection fluorescence microscopy, confocal laser scanning microscopy, and high-throughput imaging. Applying this technology, we measured the early stage mechanical forces exerted by 3T3 fibroblasts across different nanoscale features, detecting forces ranging from 12 to 56 pN. By integrating the Mechano-Cas12a Assisted Tension Sensor (MCATS) system, we achieved rapid and high-throughput quantification of cellular traction forces, analyzing over 2 million cells within minutes. Our findings reveal that nanoscale clustering of integrin ligands significantly influences the mechanical responses of cells. This platform offers a powerful tool for mechanobiology research, facilitating the study of cellular forces and mechanotransduction pathways in a high-throughput manner compatible with standard cell culture systems.
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Applied Materials & Interfaces
工程技术-材料科学:综合
CiteScore
16.00
自引率
6.30%
发文量
4978
审稿时长
1.8 months
期刊介绍:
ACS Applied Materials & Interfaces is a leading interdisciplinary journal that brings together chemists, engineers, physicists, and biologists to explore the development and utilization of newly-discovered materials and interfacial processes for specific applications. Our journal has experienced remarkable growth since its establishment in 2009, both in terms of the number of articles published and the impact of the research showcased. We are proud to foster a truly global community, with the majority of published articles originating from outside the United States, reflecting the rapid growth of applied research worldwide.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


