Rational multienzyme architecture design with iMARS
IF 42.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Biocatalytic cascades with spatial proximity can orchestrate multistep pathways to form metabolic highways, which enhance the overall catalytic efficiency. However, the effect of spatial organization on catalytic activity is poorly understood, and multienzyme architectural engineering with predictable performance remains unrealized. Here, we developed a standardized framework, called iMARS, to rapidly design the optimal multienzyme architecture by integrating high-throughput activity tests and structural analysis. The approach showed potential for industrial-scale applications, with artificial fusion enzymes designed by iMARS significantly improving the production of resveratrol by 45.1-fold and raspberry ketone by 11.3-fold in vivo, as well as enhancing ergothioneine synthesis in fed-batch fermentation. In addition, iMARS greatly enhanced the in vitro catalytic efficiency of the multienzyme complexes for PET plastic depolymerization and vanillin biosynthesis. As a generalizable and flexible strategy at molecular level, iMARS could greatly facilitate green chemistry, synthetic biology, and biomanufacturing.
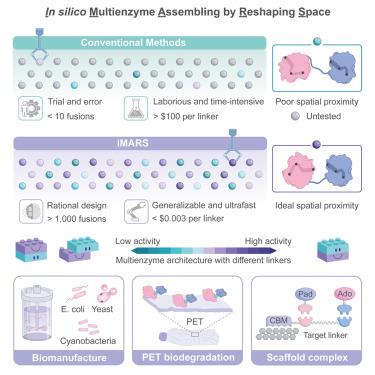
基于iMARS的合理多酶结构设计
具有空间亲和性的生物催化级联可以协调多步途径形成代谢高速公路,从而提高整体催化效率。然而,空间组织对催化活性的影响知之甚少,具有可预测性能的多酶建筑工程仍未实现。在这里,我们开发了一个标准化的框架,称为iMARS,通过集成高通量活性测试和结构分析来快速设计最佳的多酶结构。该方法具有工业规模应用的潜力,iMARS设计的人工融合酶显着提高了体内白藜芦醇的产量45.1倍,覆盆子酮的产量提高了11.3倍,并提高了补料分批发酵中麦角硫因的合成。此外,iMARS极大地提高了多酶配合物对PET塑料解聚和香兰素生物合成的体外催化效率。作为一种在分子水平上具有通用性和灵活性的策略,iMARS可以极大地促进绿色化学、合成生物学和生物制造。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
文献相关原料
公司名称
产品信息
索莱宝
PBS buffer
麦克林
Formic acid
麦克林
Tri?uoroacetic acid
麦克林
Vitamin B6
麦克林
Corn steep liquor
麦克林
Decane
麦克林
Malonic acid
麦克林
Phenylmethylsulfonyl fluoride
麦克林
Guanidine hydrochloride
麦克林
Bovine serum albumin
麦克林
Isoeugenol
麦克林
Vanillin
麦克林
Raspberry ketone
麦克林
4-Vinylguaiacol
麦克林
Terephthalic acid
麦克林
Resveratrol
麦克林
Glycerol
麦克林
ATP
麦克林
Ammonium ferrous sulfate
麦克林
L-Cysteine monohydrochloride
麦克林
L-Methionine
麦克林
L-Histidine
麦克林
L-Arabinose
麦克林
Ammonia
麦克林
Glycerol
麦克林
Iron (II) sulfate heptahydrate
麦克林
PEG 20000
麦克林
Tris-HCl
麦克林
Imidazole
麦克林
Hydrochloric acid
麦克林
Acetonitrile
麦克林
Glycine
麦克林
Glucose
麦克林
Chloramphenicol
麦克林
Magnesium sulfate heptahydrate
麦克林
Sodium phosphate dibasic
麦克林
Manganese (II) chloride
麦克林
Potassium phosphate
麦克林
Calcium chloride
麦克林
Magnesium chloride
麦克林
Sodium hydroxide
麦克林
Sodium chloride
阿拉丁
Ferulic acid
阿拉丁
p-Coumaric acid
阿拉丁
Spectinomycin
阿拉丁
Magnesium sulfate heptahydrate
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


