Redox-Neutral Photocatalytic Germylative Difunctionalization of Unactivated Olefins via Selective Radical Capture by Ge(II)
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
This study reports the development of a photocatalytic germanyl functionalization of unactivated olefins. A modular approach to alkylgermanium trihalides was realized through a three-component radical reaction involving fluoroalkyl bromides and commercially available GeBr2 in a redox-neutral process. The target organogermanes were readily obtained via one-pot derivatization using various Grignard reagents, demonstrating good functional group compatibility with amides, esters and ketones. This work highlights the utility of Ge(II) reagents in constructing C–Ge bonds via photoredox catalysis. Notably, the selectivity in capturing electrophilic or nucleophilic radicals by Ge(II) was found to be significantly influenced by the halide ligands on GeX2.
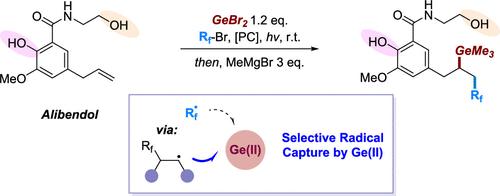
Ge(II)选择性自由基捕获下氧化还原-中性光催化非活化烯烃萌发双功能化
本研究报道了未活化烯烃的光催化锗基功能化的进展。通过在氧化还原中性过程中涉及氟烷基溴和市售GeBr2的三组分自由基反应,实现了烷基锗三卤化物的模块化方法。用各种格氏试剂进行一锅衍生反应,得到了目标有机化合物,与酰胺类、酯类和酮类具有良好的官能团相容性。这项工作强调了Ge(II)试剂在通过光氧化还原催化构建C-Ge键中的应用。值得注意的是,Ge(II)捕获亲电或亲核自由基的选择性受到GeX2上卤化物配体的显著影响。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


