Circulating tumour DNA in early stage and locally advanced NSCLC: ready for clinical implementation?
IF 82.2
1区 医学
Q1 ONCOLOGY
引用次数: 0
Abstract
Circulating tumour DNA (ctDNA) can be released by cancer cells into biological fluids through apoptosis, necrosis or active release. In patients with non-small-cell lung cancer (NSCLC), ctDNA levels correlate with clinical and pathological factors, including histology, tumour size and proliferative status. Currently, ctDNA analysis is recommended for molecular profiling in patients with advanced-stage NSCLC. In this Review, we summarize the increasing evidence suggesting that ctDNA has potential clinical applications in the management of patients with early stage and locally advanced NSCLC. In those with early stage NSCLC, detection of ctDNA before and/or after surgery is associated with a greater risk of disease recurrence. Longitudinal monitoring after surgery can further increase the prognostic value of ctDNA testing and enables detection of disease recurrence earlier than the assessment of clinical or radiological progression. In patients with locally advanced NSCLC, the detection of ctDNA after chemoradiotherapy is also associated with a greater risk of disease progression. Owing to the limited number of patients enrolled and the different technologies used for ctDNA testing in most of the clinical studies performed thus far, their results are not sufficient to currently support the routine clinical use of ctDNA monitoring in patients with early stage or locally advanced NSCLC. Therefore, we discuss the need for interventional studies to provide evidence for implementing ctDNA testing in this setting. Analysis of circulating tumour DNA (ctDNA) is commonly used for molecular profiling in patients with advanced-stage non-small-cell lung cancer (NSCLC). The authors of this Review summarize the available evidence on the potential utility of incorporating ctDNA in the management of those with early stage and locally advanced NSCLC and propose interventional studies to provide the necessary additional evidence.

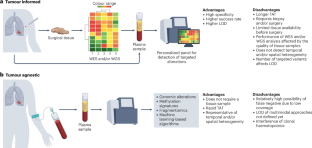
早期和局部晚期 NSCLC 中的循环肿瘤 DNA:是否已准备好临床应用?
循环肿瘤DNA (ctDNA)可由癌细胞通过凋亡、坏死或主动释放的方式释放到生物体液中。在非小细胞肺癌(NSCLC)患者中,ctDNA水平与临床和病理因素相关,包括组织学、肿瘤大小和增殖状态。目前,ctDNA分析被推荐用于晚期NSCLC患者的分子谱分析。在这篇综述中,我们总结了越来越多的证据表明ctDNA在早期和局部晚期NSCLC患者的治疗中具有潜在的临床应用。在早期非小细胞肺癌患者中,手术前和/或手术后检测ctDNA与更高的疾病复发风险相关。术后纵向监测可以进一步提高ctDNA检测的预后价值,并使疾病复发的检测早于临床或放射学进展的评估。在局部晚期NSCLC患者中,放化疗后检测ctDNA也与疾病进展的风险增加相关。由于入组的患者数量有限,并且迄今为止进行的大多数临床研究中ctDNA检测使用的技术不同,其结果目前不足以支持ctDNA监测在早期或局部晚期NSCLC患者中的常规临床应用。因此,我们讨论介入研究的必要性,为在这种情况下实施ctDNA检测提供证据。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
99.40
自引率
0.40%
发文量
114
审稿时长
6-12 weeks
期刊介绍:
Nature Reviews publishes clinical content authored by internationally renowned clinical academics and researchers, catering to readers in the medical sciences at postgraduate levels and beyond. Although targeted at practicing doctors, researchers, and academics within specific specialties, the aim is to ensure accessibility for readers across various medical disciplines. The journal features in-depth Reviews offering authoritative and current information, contextualizing topics within the history and development of a field. Perspectives, News & Views articles, and the Research Highlights section provide topical discussions, opinions, and filtered primary research from diverse medical journals.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


