Discovery of 2,4-Quinazolinedione Derivatives as LC3B Recruiters in the Facilitation of Protein Complex Degradations
IF 6
2区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
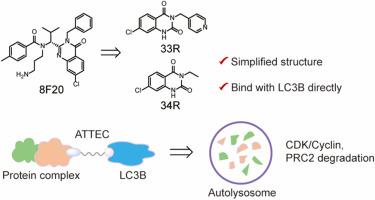
Targeted protein degradation through autophagosome-tethering compounds (ATTECs) that bypasses the ubiquitination process has garnered increasing attention. LC3B, a key protein in autophagosome formation, recruits substrates into the autophagy-lysosome system for degradation. In this study, we systematically optimized 2,4-quinazolinedione derivatives as LC3B-recruiting fragments, utilizing the CDK9 indicator. By attaching the designed LC3B-recruiting fragment to CDK9 inhibitor SNS-032 through a linker, the resulting bifunctional ATTEC molecule simultaneously degraded CDK9 and its associated Cyclin T1. Two-dimensional NMR experiments confirmed the direct interaction between the novel LC3B-recruiting fragments and LC3B. Mechanistic studies elucidated that degradation occurred via an LC3B-dependent autophagy-lysosomal pathway. Additionally, the general applicability of leveraging LC3B-recruiting fragments linked to inhibitors for the targeted degradation of protein complexes was validated with PRC2 and CDK2/4/6 along with their respective Cyclins. This work provides a series of novel LC3B-recruiting fragments that enrich the ATTEC toolbox and can be applied to the degradation of diverse intracellular disease-causing proteins.
2,4-喹唑啉二酮衍生物在促进蛋白质复合物降解中作为LC3B招募者的发现
通过自噬体系固化合物(attec)绕过泛素化过程的靶向蛋白质降解已经引起了越来越多的关注。LC3B是自噬体形成的关键蛋白,它将底物招募到自噬-溶酶体系统中进行降解。在本研究中,我们利用CDK9指标系统地优化了2,4-喹唑啉二酮衍生物作为lc3b招募片段。通过连接器将设计的lc3b招募片段连接到CDK9抑制剂sn -032上,得到的双功能ATTEC分子同时降解CDK9及其相关的Cyclin T1。二维核磁共振实验证实了新型LC3B招募片段与LC3B之间的直接相互作用。机制研究表明,降解是通过lc3b依赖性自噬-溶酶体途径发生的。此外,通过PRC2和CDK2/4/6及其各自的细胞周期蛋白,验证了利用lc3b招募片段连接抑制剂靶向降解蛋白质复合物的一般适用性。这项工作提供了一系列新的lc3b招募片段,丰富了ATTEC工具箱,可以应用于多种细胞内致病蛋白的降解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
11.70
自引率
9.00%
发文量
863
审稿时长
29 days
期刊介绍:
The European Journal of Medicinal Chemistry is a global journal that publishes studies on all aspects of medicinal chemistry. It provides a medium for publication of original papers and also welcomes critical review papers.
A typical paper would report on the organic synthesis, characterization and pharmacological evaluation of compounds. Other topics of interest are drug design, QSAR, molecular modeling, drug-receptor interactions, molecular aspects of drug metabolism, prodrug synthesis and drug targeting. The journal expects manuscripts to present the rational for a study, provide insight into the design of compounds or understanding of mechanism, or clarify the targets.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


