Biofluid-Permeable and Erosion-Resistant Wireless Neural-Electronic Interfaces for Neurohomeostasis Modulation
IF 15.8
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
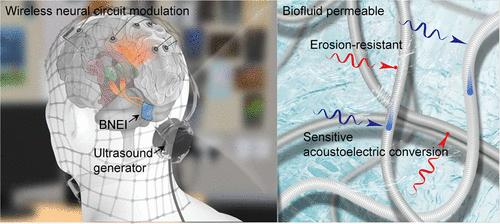
Neural-electronic interfaces through delivering electroceuticals to lesions and modulating pathological endogenous electrical environments offer exciting opportunities to treat drug-refractory neurological disorders. Such an interface should ideally be compatible with the neural tissue and aggressive biofluid environment. Unfortunately, no interface specifically designed for the biofluid environments is available so far; instead, simply stacking an encapsulation layer on silicon-based substrates makes them susceptible to biofluid leakage, device malfunction, and foreign-body reactions. Here, we developed a biofluid-permeable and erosion-resistant wireless neural-electronic interface (BNEI) that is composed of a flexible 3D interconnected poly(l-lactide) fibrous network with a dense and axially aligned piezoelectrical molecular chain arrangement architecture. The organized molecular chain structure enhances the tortuous pathway and longitudinal piezoelectric coefficient of poly(l-lactide) fibers, improves their water barrier properties, and enables efficient conversion of low-intensity acoustic vibrations transmitted in biofluids into electrical signals, achieving long-term stable and wireless neuromodulation. A 3-month clinical trial demonstrated that the BNEI can effectively accelerate the pathological cascade in peripheral neuropathy for nerve regeneration and transcranially modulate cerebellar–cerebral circuit dynamics, suppressing seizures in temporal lobe epilepsy. The BNEI can be a clinically scalable approach for wireless neuromodulation that is broadly applicable to the modulation of neurohomeostasis in both the peripheral and central nervous systems.
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Nano
工程技术-材料科学:综合
CiteScore
26.00
自引率
4.10%
发文量
1627
审稿时长
1.7 months
期刊介绍:
ACS Nano, published monthly, serves as an international forum for comprehensive articles on nanoscience and nanotechnology research at the intersections of chemistry, biology, materials science, physics, and engineering. The journal fosters communication among scientists in these communities, facilitating collaboration, new research opportunities, and advancements through discoveries. ACS Nano covers synthesis, assembly, characterization, theory, and simulation of nanostructures, nanobiotechnology, nanofabrication, methods and tools for nanoscience and nanotechnology, and self- and directed-assembly. Alongside original research articles, it offers thorough reviews, perspectives on cutting-edge research, and discussions envisioning the future of nanoscience and nanotechnology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


