mTor limits autophagy to facilitate cell volume expansion and rapid wound repair in Drosophila embryos
IF 10.7
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
Embryonic wounds repair rapidly, with no inflammation or scarring. Embryonic wound healing is driven by collective cell movements facilitated by the increase in the volume of the cells adjacent to the wound. The mechanistic target of rapamycin (mTor) complex 1 (TORC1) is associated with cell growth. We found that disrupting TORC1 signaling in Drosophila embryos prevented cell volume increases and slowed down wound repair. Catabolic processes, such as autophagy, can inhibit cell growth. Five-dimensional microscopy demonstrated that the number of autophagosomes decreased during wound repair, suggesting that autophagy must be tightly regulated for rapid wound healing. mTor inhibition increased autophagy, and activating autophagy prevented cell volume expansion and slowed down wound closure. Finally, reducing autophagy in embryos with disrupted TORC1 signaling rescued cell volume changes and rapid wound repair. Together, our results show that TORC1 activation upon wounding negatively regulates autophagy, allowing cells to increase their volumes to facilitate rapid wound healing.
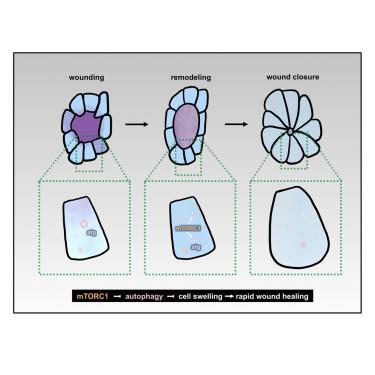
在果蝇胚胎中,mTor限制自噬以促进细胞体积扩张和快速伤口修复
胚胎伤口修复迅速,没有炎症或疤痕。胚胎伤口愈合是由靠近伤口的细胞体积增加所促进的集体细胞运动所驱动的。雷帕霉素(mTor)复合物1 (TORC1)的机制靶点与细胞生长有关。我们发现,破坏果蝇胚胎中的TORC1信号可以阻止细胞体积的增加,并减缓伤口修复。分解代谢过程,如自噬,可以抑制细胞生长。五维显微镜显示,自噬体的数量在伤口修复过程中减少,这表明自噬必须受到严格调节才能快速愈合。mTor抑制增加自噬,激活自噬阻止细胞体积扩张,减缓伤口愈合。最后,在TORC1信号中断的胚胎中,减少自噬可以挽救细胞体积变化和快速伤口修复。总之,我们的研究结果表明,损伤时TORC1的激活负调控自噬,使细胞增加体积以促进伤口快速愈合。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Developmental cell
生物-发育生物学
CiteScore
18.90
自引率
1.70%
发文量
203
审稿时长
3-6 weeks
期刊介绍:
Developmental Cell, established in 2001, is a comprehensive journal that explores a wide range of topics in cell and developmental biology. Our publication encompasses work across various disciplines within biology, with a particular emphasis on investigating the intersections between cell biology, developmental biology, and other related fields. Our primary objective is to present research conducted through a cell biological perspective, addressing the essential mechanisms governing cell function, cellular interactions, and responses to the environment. Moreover, we focus on understanding the collective behavior of cells, culminating in the formation of tissues, organs, and whole organisms, while also investigating the consequences of any malfunctions in these intricate processes.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


