Targeting FOXM1 condensates reduces breast tumour growth and metastasis
IF 48.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
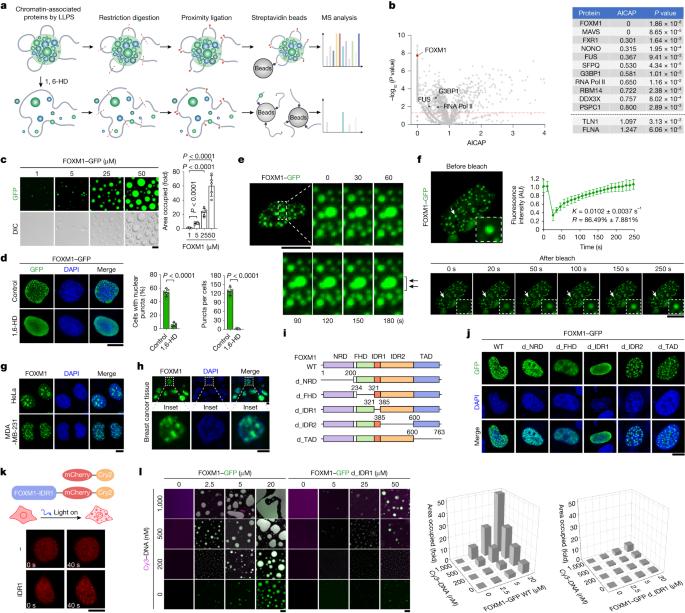
Identifying phase-separated structures remains challenging, and effective intervention methods are currently lacking1. Here we screened for phase-separated proteins in breast tumour cells and identified forkhead (FKH) box protein M1 (FOXM1) as the most prominent candidate. Oncogenic FOXM1 underwent liquid–liquid phase separation (LLPS) with FKH consensus DNA element, and compartmentalized the transcription apparatus in the nucleus, thereby sustaining chromatin accessibility and super-enhancer landscapes crucial for tumour metastatic outgrowth. Screening an epigenetics compound library identified AMPK agonists as suppressors of FOXM1 condensation. AMPK phosphorylated FOXM1 in the intrinsically disordered region (IDR), perturbing condensates, reducing oncogenic transcription, accumulating double-stranded DNA to stimulate innate immune responses, and endowing discrete FOXM1 with the ability to activate immunogenicity-related gene expressions. By developing a genetic code-expansion orthogonal system, we demonstrated that a phosphoryl moiety at a specific IDR1 site causes electrostatic repulsion, thereby abolishing FOXM1 LLPS and aggregation. A peptide targeting IDR1 and carrying the AMPK-phosphorylated residue was designed to disrupt FOXM1 LLPS and was shown to inhibit tumour malignancy, rescue tumour immunogenicity and improve tumour immunotherapy. Together, these findings provide novel and in-depth insights on function and mechanism of FOXM1 and develop methodologies that hold promising implications in clinics. The transcription factor FOXM1 forms functional condensates, the formation of which can be targeted with a specific peptide to suppress breast cancer growth and metastasis.
靶向 FOXM1 凝聚物可减少乳腺肿瘤的生长和转移
识别相分离结构仍然具有挑战性,目前缺乏有效的干预方法1。在这里,我们筛选了乳腺肿瘤细胞中的相分离蛋白,并确定叉头(FKH)盒蛋白M1 (FOXM1)是最突出的候选蛋白。致癌FOXM1与FKH共识DNA元件进行了液-液相分离(LLPS),并将细胞核中的转录装置区隔化,从而维持了染色质可及性和对肿瘤转移产物至关重要的超增强子结构。筛选表观遗传学化合物文库,确定AMPK激动剂是FOXM1缩合的抑制因子。AMPK在内在无序区(IDR)磷酸化FOXM1,扰乱凝聚物,减少致癌转录,积累双链DNA以刺激先天免疫反应,并赋予离散FOXM1激活免疫原性相关基因表达的能力。通过开发遗传密码扩展正交系统,我们证明了IDR1特定位点的磷酸化片段引起静电排斥,从而消除FOXM1的LLPS和聚集。一种靶向IDR1并携带ampk磷酸化残基的肽被设计用于破坏FOXM1 LLPS,并被证明可以抑制肿瘤恶性,挽救肿瘤免疫原性并改善肿瘤免疫治疗。总之,这些发现为FOXM1的功能和机制提供了新颖而深入的见解,并开发了具有临床应用前景的方法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


