Can a Simple Surrogate Model System Be Used to Develop a Continuous Flow Packed Bed Hydrogenation for a Complex Molecule?
IF 3.1
3区 化学
Q2 CHEMISTRY, APPLIED
引用次数: 0
Abstract
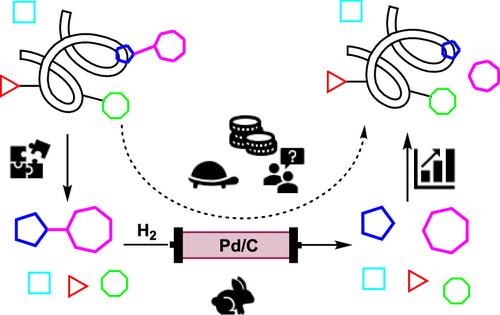
Catalytic hydrogenations are key processes in the fine chemical and pharmaceutical industries, but the development of such processes is challenging due to aspects such as catalyst deactivation, metal leaching, mass transfer limitations, solubility issues, and the formation of side products. Processes are particularly difficult to develop when a substrate is a large molecule containing multiple functional groups. These difficulties are significant obstacles for the identification of robust operating conditions; thus, workflows are necessary to speed up development timelines. The use of a more cost-effective and commercially available surrogate in development is an alternative strategy to find the optimized conditions, which can then be subsequently validated on the real molecule only at a later stage in development. The approach we apply herein is designed to use less of the real compound while minimizing the perceived risk of failure when transferring the conditions to the complex molecule. In this article, we apply our workflow for the catalytic hydrogenolysis of a large glycopeptide molecule, Cbz-protected glycopeptide (Cbz-GP), in a packed bed reactor. As part of the workflow, we use a robustness screening approach, introduced by Collins and Glorius, to show that a surrogate molecule, Cbz-protected lysine (Cbz-Lys), in the presence of additives can mimic secondary functional groups present in Cbz-GP or represent residual impurities generated upstream in the synthesis of Cbz-GP. The data generated for Cbz-Lys enabled the identification of the operating conditions for the successful deprotection of Cbz-GP after minor modification. Gratifyingly, only a few additional experiments were necessary using the Cbz-protected GP molecule to modify the conditions to achieve >95% conversion under mild conditions and within <10 s of contact time for stable performance over >6 h operation time.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
6.90
自引率
14.70%
发文量
251
审稿时长
2 months
期刊介绍:
The journal Organic Process Research & Development serves as a communication tool between industrial chemists and chemists working in universities and research institutes. As such, it reports original work from the broad field of industrial process chemistry but also presents academic results that are relevant, or potentially relevant, to industrial applications. Process chemistry is the science that enables the safe, environmentally benign and ultimately economical manufacturing of organic compounds that are required in larger amounts to help address the needs of society. Consequently, the Journal encompasses every aspect of organic chemistry, including all aspects of catalysis, synthetic methodology development and synthetic strategy exploration, but also includes aspects from analytical and solid-state chemistry and chemical engineering, such as work-up tools,process safety, or flow-chemistry. The goal of development and optimization of chemical reactions and processes is their transfer to a larger scale; original work describing such studies and the actual implementation on scale is highly relevant to the journal. However, studies on new developments from either industry, research institutes or academia that have not yet been demonstrated on scale, but where an industrial utility can be expected and where the study has addressed important prerequisites for a scale-up and has given confidence into the reliability and practicality of the chemistry, also serve the mission of OPR&D as a communication tool between the different contributors to the field.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


