Induction of Antigen-Specific Tolerance in a Multiple Sclerosis Model without Broad Immunosuppression
IF 15.8
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
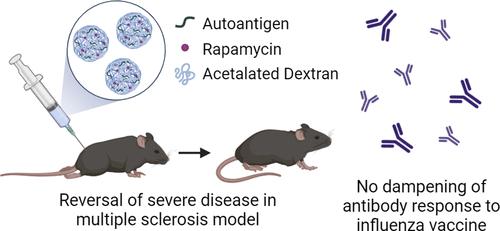
Multiple sclerosis (MS) is a severe autoimmune disorder that wreaks havoc on the central nervous system, leading to a spectrum of motor and cognitive impairments. There is no cure, and current treatment strategies rely on broad immunosuppression, leaving patients vulnerable to infections. To address this problem, our approach aims to induce antigen-specific tolerance, a much-needed shift in MS therapy. We have engineered a tolerogenic therapy consisting of spray-dried particles made of a degradable biopolymer, acetalated dextran, and loaded with an antigenic peptide and tolerizing drug, rapamycin (Rapa). After initial characterization and optimization, particles were tested in a myelin oligodendrocyte glycoprotein (MOG)-induced experimental autoimmune encephalomyelitis model of MS. Representing the earliest possible time of diagnosis, mice were treated at symptom onset in an early therapeutic model, where particles containing MOG and particles containing Rapa+MOG evoked significant reductions in clinical score. Particles were then applied to a highly clinically relevant late therapeutic model during peak disease, where MOG particles and Rapa+MOG particles each elicited a dramatic therapeutic effect, reversing hind limb paralysis and restoring fully functional limbs. To confirm the antigen specificity of our therapy, we immunized mice against the influenza antigen hemagglutinin (HA) and treated them with MOG particles or Rapa+MOG particles. The particles did not suppress antibody responses against HA. Our findings underscore the potential of this particle-based therapy to reverse autoimmunity in disease-relevant models without compromising immune competence, setting it apart from existing treatments.
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Nano
工程技术-材料科学:综合
CiteScore
26.00
自引率
4.10%
发文量
1627
审稿时长
1.7 months
期刊介绍:
ACS Nano, published monthly, serves as an international forum for comprehensive articles on nanoscience and nanotechnology research at the intersections of chemistry, biology, materials science, physics, and engineering. The journal fosters communication among scientists in these communities, facilitating collaboration, new research opportunities, and advancements through discoveries. ACS Nano covers synthesis, assembly, characterization, theory, and simulation of nanostructures, nanobiotechnology, nanofabrication, methods and tools for nanoscience and nanotechnology, and self- and directed-assembly. Alongside original research articles, it offers thorough reviews, perspectives on cutting-edge research, and discussions envisioning the future of nanoscience and nanotechnology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


