Dynamic PRC1–CBX8 stabilizes a porous structure of chromatin condensates
IF 12.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
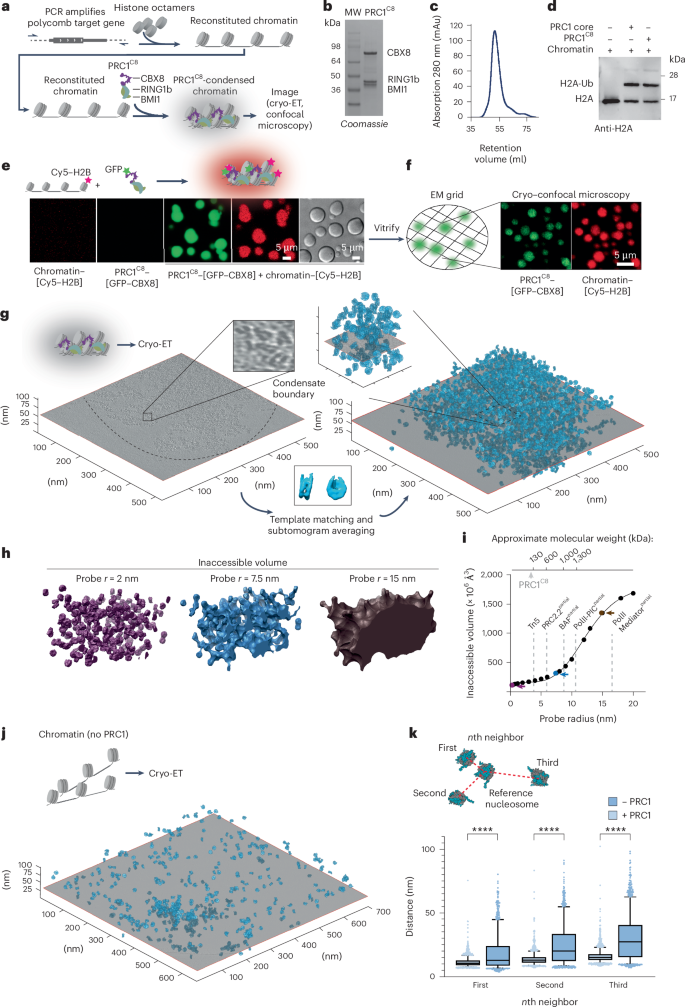
The compaction of chromatin is a prevalent paradigm in gene repression. Chromatin compaction is commonly thought to repress transcription by restricting chromatin accessibility. However, the spatial organization and dynamics of chromatin compacted by gene-repressing factors are unknown. Here, using cryo-electron tomography, we solved the three-dimensional structure of chromatin condensed by the polycomb repressive complex 1 (PRC1) in a complex with CBX8. PRC1-condensed chromatin is porous and stabilized through multivalent dynamic interactions of PRC1 with chromatin. Mechanistically, positively charged residues on the internally disordered regions of CBX8 mask negative charges on the DNA to stabilize the condensed state of chromatin. Within condensates, PRC1 remains dynamic while maintaining a static chromatin structure. In differentiated mouse embryonic stem cells, CBX8-bound chromatin remains accessible. These findings challenge the idea of rigidly compacted polycomb domains and instead provide a mechanistic framework for dynamic and accessible PRC1–chromatin condensates. Here the authors show that a gene-inactivating protein complex packs inactive genes into a dynamic and accessible structure. The study challenges the traditional views that restricted accessibility and low dynamics cause gene repression.

动态PRC1-CBX8稳定了染色质凝聚物的多孔结构
染色质的压实是基因抑制的一个普遍范例。染色质压实通常被认为是通过限制染色质的可及性来抑制转录。然而,染色质的空间组织和动态压缩的基因抑制因子是未知的。在这里,我们使用低温电子断层扫描,解决了染色质由多梳抑制复合体1 (PRC1)与CBX8络合物凝聚的三维结构。通过PRC1与染色质的多价动态相互作用,PRC1浓缩染色质具有多孔性和稳定性。机制上,CBX8内部无序区域上带正电荷的残基掩盖了DNA上的负电荷,以稳定染色质的凝聚状态。在凝析物中,PRC1在保持静态染色质结构的同时保持动态。在分化的小鼠胚胎干细胞中,cbx8结合的染色质仍然是可接近的。这些发现挑战了刚性紧致多梳结构域的观点,并为动态和可接近的prc1 -染色质凝聚物提供了一个机制框架。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


