Tumor Vaccine Exploiting Membranes with Influenza Virus-Induced Immunogenic Cell Death to Decorate Polylactic Coglycolic Acid Nanoparticles
IF 15.8
1区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
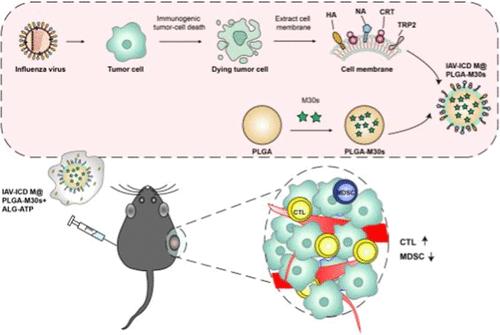
Immunogenic cell death (ICD) of tumor cells, which is characterized by releasing immunostimulatory “find me” and “eat me” signals, expressing proinflammatory cytokines and providing personalized and broad-spectrum tumor antigens draws increasing attention in developing a tumor vaccine. In this study, we aimed to investigate whether the influenza virus (IAV) is efficient enough to induce ICD in tumor cells and an extra modification of IAV components such as hemeagglutinin (HA) will be helpful for the ICD-induced cells to elicit robust antitumor effects; in addition, to evaluate whether the membrane-engineering polylactic coglycolic acid nanoparticles (PLGA NPs) simulating ICD immune stimulation mechanisms hold the potential to be a promising vaccine candidate, a mouse melanoma cell line (B16–F10 cell) was infected with IAV rescued by the reverse genetic system, and the prepared cells and membrane-modified PLGA NPs were used separately to immunize the melanoma-bearing mice. IAV-infected tumor cells exhibit dying status, releasing high mobility group box-1 (HMGB1) and adenosine triphosphate (ATP), and exposing calreticulin (CRT), IAV hemeagglutinin (HA), and tumor antigens like tyrosinase-related protein 2 (TRP2). IAV-induced ICD cells enhance biomass-derived carbon (BMDCs) migration, antigen uptake, cross-presentation, and maturation in vitro. Furthermore, immunization with IAV-induced ICD cells effectively suppressed tumor growth in melanoma-bearing mice. The isolated cell membrane inherited the immunological characteristics from the ICD cells and elicited robust antitumor immune responses through decorating PLGA NPs loading with a tumor-specific helper T-cell peptide and supplemented with ATP in a hydrogel system. This study indicated a promising strategy for developing cell-based and personalized tumor vaccines through fully taking advantage of the immune stimulation mechanisms of ICD occurrence in tumor cells, IAV modification, and nanoscale delivery.
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Nano
工程技术-材料科学:综合
CiteScore
26.00
自引率
4.10%
发文量
1627
审稿时长
1.7 months
期刊介绍:
ACS Nano, published monthly, serves as an international forum for comprehensive articles on nanoscience and nanotechnology research at the intersections of chemistry, biology, materials science, physics, and engineering. The journal fosters communication among scientists in these communities, facilitating collaboration, new research opportunities, and advancements through discoveries. ACS Nano covers synthesis, assembly, characterization, theory, and simulation of nanostructures, nanobiotechnology, nanofabrication, methods and tools for nanoscience and nanotechnology, and self- and directed-assembly. Alongside original research articles, it offers thorough reviews, perspectives on cutting-edge research, and discussions envisioning the future of nanoscience and nanotechnology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


