Pd-Catalyzed Dehydrogenation Enhanced by Charge Transfer from MoOx Promoter
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
Incorporating metal oxides is a sensible strategy for enhancing the efficiency of precious metals. Upon the introduction of molybdenum oxide, diverse coordination structures were formed on the support surface depending on the concentration used. This modified surface architecture orchestrates consequential alterations in the electronic and geometric configurations of the active metal, concurrently influencing the catalytic performance. In this study, MoOx species were introduced into Pd in a controlled manner to substantially enhance the dehydrogenation activity of the N-heterocyclic liquid organic hydrogen carrier system. Pd−MoOx/Al2O3 catalyst─featuring an optimal 0.18 wt % Mo loading─demonstrated noteworthy improvement in activity, surpassing Pd/Al2O3 by factors of 1.57 and 1.24, accompanied by enhanced recyclability in two distinct LOHC systems. Integrated characterization and theoretical calculations elucidated the modification of the electronic properties of both Pd and Al2O3 upon MoOx introduction and the corresponding adsorption behavior of the reactants, highlighting the charge transfer phenomenon from Pd to MoOx.
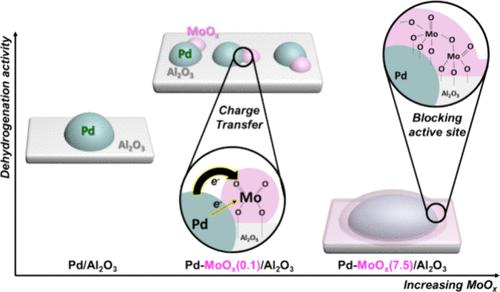
通过氧化钼促进剂的电荷转移增强钯催化脱氢反应
加入金属氧化物是提高贵金属效率的明智策略。在引入氧化钼后,根据使用的浓度不同,在支撑表面形成了不同的配位结构。这种修饰的表面结构协调了活性金属的电子和几何构型的相应改变,同时影响了催化性能。本研究将MoOx物种可控地引入Pd中,大大提高了n -杂环液态有机氢载体体系的脱氢活性。Pd−MoOx/Al2O3催化剂的最佳Mo负载为0.18 wt %,其活性显著提高,超过Pd/Al2O3的1.57和1.24倍,同时在两种不同的LOHC体系中可回收性增强。综合表征和理论计算阐明了引入MoOx后Pd和Al2O3的电子性质的改变以及相应的吸附行为,突出了Pd到MoOx的电荷转移现象。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


