Empowering Sustainable Energy: Lead-Coated Plastic Chip Electrodes for Effective CO2 Reduction
IF 3.7
2区 化学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Electrochemical CO2 reduction is crucial in combatting climate change and advancing sustainable energy practices by converting CO2 into valuable chemicals and fuels, thereby reducing atmospheric CO2 levels and enabling the storage and utilization of renewable energy from intermittent sources like solar and wind. The selection of electrode materials and platform design plays a critical role in enhancing reaction efficiency and product selectivity during CO2 reduction. Various metals, both in their solid forms and coated over substrates, have been used in electrochemical CO2RR. In this study, we utilized electrodeposition to modify the plastic chip electrode (PCE), depositing lead metal onto it through a galvanostatic method at a current density of 100 mA/cm2 from a 0.1 M Pb(NO3)2 aqueous solution. Pb-coated electrodes are crucial due to their high selectivity, efficiency, cost-effectiveness, and flexibility as electrode materials. Their good stability and durability make them ideal for long-term applications. The electrochemical reduction of carbon dioxide using the Pb/PCE electrode as the cathode has been investigated, focusing on assessing how different electrolysis potentials influenced the faradaic efficiency of formic acid production. Our results demonstrated that the peak faradaic efficiency, reaching 86.2%, was achieved at −0.7 V vs RHE over a 5 h electrolysis period.
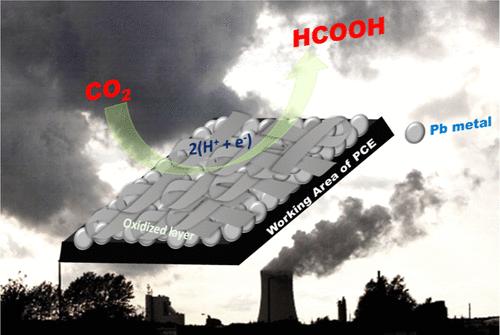
授权可持续能源:有效减少二氧化碳的铅涂覆塑料芯片电极
通过将二氧化碳转化为有价值的化学品和燃料,从而降低大气中的二氧化碳水平,并实现太阳能和风能等间歇性可再生能源的储存和利用,电化学二氧化碳减排对于应对气候变化和推进可持续能源实践至关重要。在CO2还原过程中,电极材料的选择和平台的设计对提高反应效率和产物选择性起着至关重要的作用。各种金属,无论是固体形式还是涂覆在衬底上,都已用于电化学CO2RR。在这项研究中,我们利用电沉积技术对塑料芯片电极(PCE)进行了修饰,在0.1 M的Pb(NO3)2水溶液中,以100 mA/cm2的电流密度通过恒流法将金属铅沉积在PCE上。由于其作为电极材料的高选择性、高效率、成本效益和灵活性,铅涂覆电极是至关重要的。其良好的稳定性和耐用性使其成为长期应用的理想选择。研究了Pb/PCE电极作为阴极对二氧化碳的电化学还原,重点评估了不同的电解电位对甲酸生产的法拉第效率的影响。结果表明,在−0.7 V vs RHE条件下,在5 h的电解时间内,法拉第效率达到了86.2%的峰值。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Langmuir
化学-材料科学:综合
CiteScore
6.50
自引率
10.30%
发文量
1464
审稿时长
2.1 months
期刊介绍:
Langmuir is an interdisciplinary journal publishing articles in the following subject categories:
Colloids: surfactants and self-assembly, dispersions, emulsions, foams
Interfaces: adsorption, reactions, films, forces
Biological Interfaces: biocolloids, biomolecular and biomimetic materials
Materials: nano- and mesostructured materials, polymers, gels, liquid crystals
Electrochemistry: interfacial charge transfer, charge transport, electrocatalysis, electrokinetic phenomena, bioelectrochemistry
Devices and Applications: sensors, fluidics, patterning, catalysis, photonic crystals
However, when high-impact, original work is submitted that does not fit within the above categories, decisions to accept or decline such papers will be based on one criteria: What Would Irving Do?
Langmuir ranks #2 in citations out of 136 journals in the category of Physical Chemistry with 113,157 total citations. The journal received an Impact Factor of 4.384*.
This journal is also indexed in the categories of Materials Science (ranked #1) and Multidisciplinary Chemistry (ranked #5).
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


