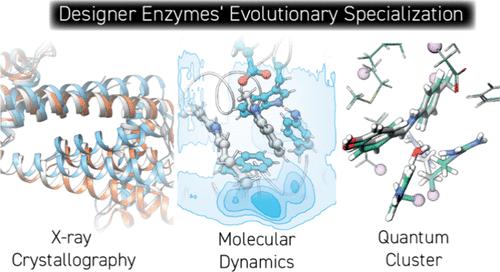
Evolutionary Specialization of a Promiscuous Designer Enzyme
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
The evolution of a promiscuous enzyme for its various activities often results in catalytically specialized variants. This is an important natural mechanism to ensure the proper functioning of natural metabolic networks. It also acts as both a curse and blessing for enzyme engineers, where enzymes that have undergone directed evolution may exhibit exquisite selectivity at the expense of a diminished overall catalytic repertoire. We previously performed two independent directed evolution campaigns on a promiscuous designer enzyme that leverages the unique properties of a noncanonical amino acid (ncAA) para-aminophenylalanine (pAF) as catalytic residue, resulting in two evolved variants which are both catalytically specialized. Here, we combine mutagenesis, crystallography, and computation to reveal the molecular basis of the specialization phenomenon. In one evolved variant, an unexpected change in quaternary structure biases substrate dynamics to promote enantioselective catalysis, while the other demonstrates synergistic cooperation between natural side chains and the pAF residue to form semisynthetic catalytic machinery.
混杂设计酶的进化专门化
一种具有多种活性的杂化酶在进化过程中往往会产生催化特化的变体。这是确保自然代谢网络正常运作的重要自然机制。对于酶工程学家来说,这既是祸也是福,经历了定向进化的酶可能会表现出精湛的选择性,但代价是总体催化范围的缩小。我们以前曾对一种杂合设计酶进行过两次独立的定向进化,该酶利用了一种非典型氨基酸(ncAA)对氨基苯丙氨酸(pAF)作为催化残基的独特性质,从而产生了两种在催化上都很特化的进化变体。在这里,我们将诱变、晶体学和计算结合起来,揭示了特化现象的分子基础。在其中一个进化变体中,四元结构的意外变化使底物动力学发生偏移,从而促进了对映体选择性催化,而另一个进化变体则展示了天然侧链与 pAF 残基之间的协同合作,从而形成了半合成催化机制。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


