Organic electrolyte cations promote non-aqueous CO2 reduction by mediating interfacial electric fields
IF 42.8
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
The electrochemical reduction of CO2 is sensitive to the microenvironment surrounding catalytic active sites. Although the impact of changing electrolyte composition on rates has been studied intensively in aqueous electrolytes, less is known about the influence of the electrochemical environment in non-aqueous solvents. Here we demonstrate that organic alkylammonium cations influence catalytic performance in non-aqueous media and describe a physical model that rationalizes these observations. Using results from kinetic, spectroscopic and computational techniques, we argue that the strength of the electric field at the catalyst surface is sensitive to the molecular identity of the organic cation in the electrolyte. This is true irrespective of solvent, electrolyte ionic strength or electrolyte anion. Our results suggest that changes in the interfacial electric field strength can be attributed to differences in the cation–electrode distance. Changes in the electric field strength affect CO formation rates as they modify the energetics of the kinetically relevant CO2 activation step. Electrolyte cations have been shown to have a strong impact on reactivity in electrocatalytic CO2 reduction. However, most studies have been performed in an aqueous environment. Here the effect of various alkylammonium cations on CO2 reduction in aprotic solvents is investigated, with the interfacial electric field induced by the cations shown to be a dominant factor.

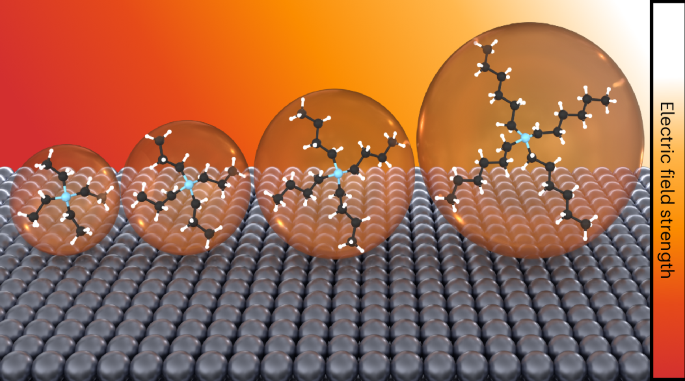
有机电解质阳离子通过介面电场促进非水CO2还原
CO2的电化学还原对催化活性位点周围的微环境很敏感。虽然电解质成分的变化对水性电解质速率的影响已经得到了深入的研究,但对非水性溶剂中电化学环境的影响知之甚少。在这里,我们证明了有机烷基铵阳离子在非水介质中影响催化性能,并描述了一个物理模型,使这些观察结果合理化。利用动力学、光谱学和计算技术的结果,我们认为催化剂表面的电场强度对电解质中有机阳离子的分子特性很敏感。无论溶剂、电解质离子强度或电解质阴离子强度如何,这都是正确的。我们的研究结果表明,界面电场强度的变化可归因于阳离子-电极距离的差异。电场强度的变化影响CO的生成速率,因为它们改变了与动力学相关的CO2激活步骤的能量学。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Catalysis
Chemical Engineering-Bioengineering
CiteScore
52.10
自引率
1.10%
发文量
140
期刊介绍:
Nature Catalysis serves as a platform for researchers across chemistry and related fields, focusing on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, encompassing both fundamental and applied studies. With a particular emphasis on advancing sustainable industries and processes, the journal provides comprehensive coverage of catalysis research, appealing to scientists, engineers, and researchers in academia and industry.
Maintaining the high standards of the Nature brand, Nature Catalysis boasts a dedicated team of professional editors, rigorous peer-review processes, and swift publication times, ensuring editorial independence and quality. The journal publishes work spanning heterogeneous catalysis, homogeneous catalysis, and biocatalysis, covering areas such as catalytic synthesis, mechanisms, characterization, computational studies, nanoparticle catalysis, electrocatalysis, photocatalysis, environmental catalysis, asymmetric catalysis, and various forms of organocatalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


