A BCG kill switch strain protects against Mycobacterium tuberculosis in mice and non-human primates with improved safety and immunogenicity
IF 20.5
1区 生物学
Q1 MICROBIOLOGY
引用次数: 0
Abstract
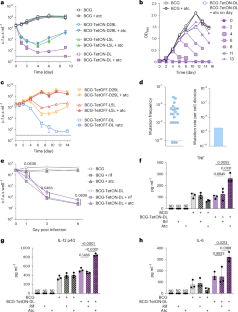
Improved vaccination strategies for tuberculosis are needed. Intravenous (i.v.) delivery of live attenuated Mycobacterium bovis BCG provides protection against Mycobacterium tuberculosis (Mtb) in macaques but poses safety challenges. Here we genetically engineered two strains, BCG-TetON-DL and BCG-TetOFF-DL, to either induce or inhibit expression of two phage lysin operons, respectively, upon tetracycline exposure. We show that lysin expression kills BCG in vitro, in infected macrophages, and following infection of immunocompetent (C57BL/6) and immunocompromised (SCID) mice. Modified BCG elicited similar immune responses and provided similar protection against Mtb challenge as wild-type BCG in mice. In macaques, cessation of tetracycline treatment reduced i.v.-administered BCG-TetOFF-DL numbers. Intravenous BCG-TetOFF-DL increased pulmonary CD4 T-cell responses compared with wild-type BCG-induced responses and provided robust protection against Mtb challenge. Sterilizing immunity occurred in 6 of 8 macaques compared with 2 of 8 wild-type BCG-immunized macaques. Thus, a ‘kill-switch’ BCG strain provides additional safety and robust protection against Mtb infection. Engineered Mycobacterium bovis BCG encoding tetracycline-controlled phage lysin kill switches elicits protective immunity against subsequent M. tuberculosis infection in mice and non-human primates.

一种卡介苗杀伤开关菌株在小鼠和非人类灵长类动物中具有抗结核分枝杆菌的保护作用,提高了安全性和免疫原性
需要改进结核病疫苗接种战略。静脉注射减毒牛分枝杆菌卡介苗可保护猕猴免受结核分枝杆菌(Mtb)的感染,但存在安全性问题。在这里,我们通过基因工程改造了两株菌株,BCG-TetON-DL和BCG-TetOFF-DL,分别诱导或抑制四环素暴露后两种噬菌体溶酶操纵子的表达。我们发现,在体外、感染巨噬细胞以及感染免疫正常(C57BL/6)和免疫功能低下(SCID)小鼠后,溶酶表达可杀死BCG。改良卡介苗在小鼠体内引起了与野生型卡介苗相似的免疫应答,并提供了与野生型卡介苗相似的抗结核攻击保护。在猕猴中,停止四环素治疗可降低静脉注射BCG-TetOFF-DL的数量。与野生型bcg诱导的应答相比,静脉注射BCG-TetOFF-DL增加了肺部CD4 t细胞应答,并提供了对Mtb攻击的强大保护。8只猕猴中有6只出现绝育免疫,而8只野生型bcg免疫猕猴中只有2只出现绝育免疫。因此,“杀伤开关”卡介苗毒株提供了额外的安全性和针对结核分枝杆菌感染的强大保护。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Microbiology
Immunology and Microbiology-Microbiology
CiteScore
44.40
自引率
1.10%
发文量
226
期刊介绍:
Nature Microbiology aims to cover a comprehensive range of topics related to microorganisms. This includes:
Evolution: The journal is interested in exploring the evolutionary aspects of microorganisms. This may include research on their genetic diversity, adaptation, and speciation over time.
Physiology and cell biology: Nature Microbiology seeks to understand the functions and characteristics of microorganisms at the cellular and physiological levels. This may involve studying their metabolism, growth patterns, and cellular processes.
Interactions: The journal focuses on the interactions microorganisms have with each other, as well as their interactions with hosts or the environment. This encompasses investigations into microbial communities, symbiotic relationships, and microbial responses to different environments.
Societal significance: Nature Microbiology recognizes the societal impact of microorganisms and welcomes studies that explore their practical applications. This may include research on microbial diseases, biotechnology, or environmental remediation.
In summary, Nature Microbiology is interested in research related to the evolution, physiology and cell biology of microorganisms, their interactions, and their societal relevance.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


