Impact of RSVpreF vaccination on reducing the burden of respiratory syncytial virus in infants and older adults
IF 58.7
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
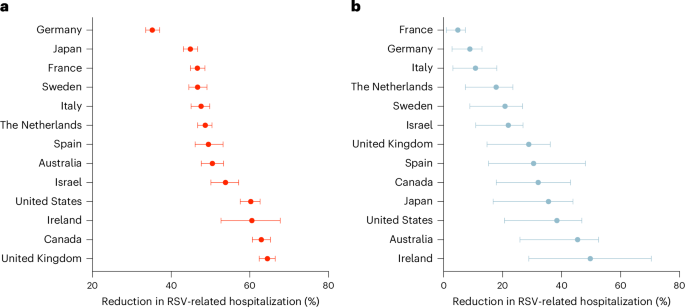
Respiratory syncytial virus (RSV) causes a substantial health burden among infants and older adults. Prefusion F protein-based vaccines have shown high efficacy against RSV disease in clinical trials, offering promise for mitigating this burden through maternal and older adult immunization. Employing an individual-based model, we evaluated the impact of RSV vaccination on hospitalizations and deaths in 13 high-income countries, assuming that the vaccine does not prevent infection or transmission. Using country-specific vaccine uptake rates for seasonal influenza, we found that vaccination of older adults would prevent hospitalizations by a median of 35–64% across the countries studied here. Vaccination of pregnant women could avert infant hospitalizations by 5–50%. Reductions in RSV-related mortality mirrored those estimated for hospitalizations. While substantial hospitalization costs could be averted, the impact of vaccination depends critically on uptake rates. Enhancing uptake and accessibility is crucial for maximizing the real-world impact of vaccination on reducing RSV burden among vulnerable populations. An individual-based mathematical model evaluating the public health benefits of RSVpreF vaccination showed that vaccinating older adults and pregnant women can substantially reduce respiratory syncytial virus (RSV)-related hospitalizations and deaths, averting substantial inpatient costs, with the impact varying across countries and influenced by both uptake rates and the local epidemiological characteristics of RSV seasons.

RSVpreF疫苗接种对减轻婴儿和老年人呼吸道合胞病毒负担的影响
呼吸道合胞病毒(RSV)在婴儿和老年人中造成巨大的健康负担。基于预融合F蛋白的疫苗在临床试验中显示出对RSV疾病的高疗效,为通过孕产妇和老年人免疫减轻这一负担提供了希望。采用基于个体的模型,我们评估了13个高收入国家RSV疫苗接种对住院和死亡的影响,假设疫苗不能预防感染或传播。使用季节性流感的国家特异性疫苗接种率,我们发现,在研究的国家中,老年人接种疫苗可预防住院的中位数为35-64%。孕妇接种疫苗可使婴儿住院率降低5-50%。rsv相关死亡率的降低反映了住院死亡率的估计。虽然可以避免大量住院费用,但疫苗接种的影响主要取决于接种率。加强疫苗的吸收和可及性对于最大限度地发挥疫苗接种对减少弱势人群呼吸道合胞病毒负担的实际影响至关重要。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


