Electrocatalytic Enantioselective Tandem C–H Indolization toward Biindolyl Atropisomers: Reaction Development and Mechanistic Insight
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
The electrochemical methods for asymmetric indolization are still elusive and pose a significant challenge. Taking advantage of 2-alkynylanilines as electrochemically compatible indolyl equivalents, we herein represent a mild and highly enantioselective electrocatalytic process for tandem C–H indolization to form 2,3′-biindolyl atropisomers along with hydrogen evolution reaction. Integrated experimental and computational mechanistic studies revealed that a sequential C–H metalation/nucleophilic cyclization/reductive elimination/anodic oxidation sequence involving a RhIII–RhI–RhIII catalytic cycle accounts for the established transformation.
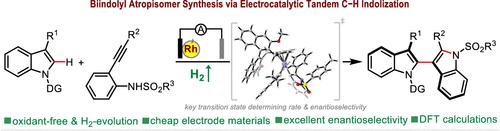
电催化对映选择性串联C-H吲哚缩二聚体:反应发展及其机理
电化学方法的不对称注射仍然是难以捉摸的,并提出了重大的挑战。利用2-炔基苯胺作为电化学相容的吲哚基等价物,我们在此提出了一种温和且高度对映选择性的电催化过程,用于串联C-H吲哚形成2,3 ' -双吲哚基托普二聚体并伴有析氢反应。综合实验和计算机制研究表明,C-H金属化/亲核环化/还原消除/阳极氧化序列涉及rhiii - rhhi - rhiii催化循环,可以解释所建立的转化。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


