The nuclear matrix stabilizes primed-specific genes in human pluripotent stem cells
IF 17.3
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
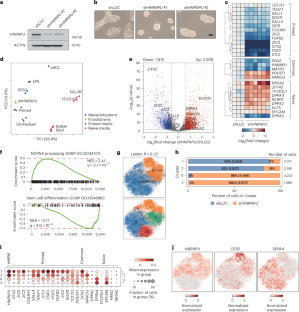
The nuclear matrix, a proteinaceous gel composed of proteins and RNA, is an important nuclear structure that supports chromatin architecture, but its role in human pluripotent stem cells (hPSCs) has not been described. Here we show that by disrupting heterogeneous nuclear ribonucleoprotein U (HNRNPU) or the nuclear matrix protein, Matrin-3, primed hPSCs adopted features of the naive pluripotent state, including morphology and upregulation of naive-specific marker genes. We demonstrate that HNRNPU depletion leads to increased chromatin accessibility, reduced DNA contacts and increased nuclear size. Mechanistically, HNRNPU acts as a transcriptional co-factor that anchors promoters of primed-specific genes to the nuclear matrix with POLII to promote their expression and their RNA stability. Overall, HNRNPU promotes cell-type stability and when reduced promotes conversion to earlier embryonic states. Ma et al. show that heterogeneous nuclear ribonucleoprotein U promotes the primed state in human pluripotent stem cells by interacting with nuclear matrix protein, Matrin-3, and regulating primed-specific genes.

核基质稳定了人类多能干细胞中的引物特异性基因
核基质是一种由蛋白质和RNA组成的蛋白凝胶,是支持染色质结构的重要核结构,但其在人类多能干细胞(hPSCs)中的作用尚未被描述。本研究表明,通过破坏异质核核糖核蛋白U (HNRNPU)或核基质蛋白Matrin-3,引物的人乳头状细胞具有幼稚多能状态的特征,包括形态和幼稚特异性标记基因的上调。我们证明HNRNPU耗损导致染色质可及性增加、DNA接触减少和核大小增加。从机制上讲,HNRNPU作为一种转录辅助因子,将引物特异性基因的启动子锚定在具有POLII的核基质上,以促进它们的表达和RNA稳定性。总的来说,HNRNPU促进细胞类型的稳定性,当减少时促进向早期胚胎状态的转化。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Nature Cell Biology
生物-细胞生物学
CiteScore
28.40
自引率
0.90%
发文量
219
审稿时长
3 months
期刊介绍:
Nature Cell Biology, a prestigious journal, upholds a commitment to publishing papers of the highest quality across all areas of cell biology, with a particular focus on elucidating mechanisms underlying fundamental cell biological processes. The journal's broad scope encompasses various areas of interest, including but not limited to:
-Autophagy
-Cancer biology
-Cell adhesion and migration
-Cell cycle and growth
-Cell death
-Chromatin and epigenetics
-Cytoskeletal dynamics
-Developmental biology
-DNA replication and repair
-Mechanisms of human disease
-Mechanobiology
-Membrane traffic and dynamics
-Metabolism
-Nuclear organization and dynamics
-Organelle biology
-Proteolysis and quality control
-RNA biology
-Signal transduction
-Stem cell biology
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


